Introduction
Activities aimed at patient safety using medical technology are becoming increasingly important. Consequently, high-use clinical diagnostic equipment such as sphygmomanometers, which provide relevant information for decision-making in the medical care part, should be taken as objects of primary care study [1]. Thus, different National Institutes of Metrology have increased studies on the application of metrology in health through legal metrological control.
The Institute of Metrology of Bosnia and Herzegovina (IMBIH) [2] highlighted the need to introduce metrology in clinical medicine, particularly in the standardization of norms associated with the inspection of medical devices, which provides a basis for the introduction of these devices in the legal metrology system and allows the measurement units to be linked with a precise definition in terms of ranges and errors. However, they did not report a deep and specific approach to estimate uncertainty for the particular case of medical equipment. Likewise, a study in Portugal discussed the role of metrology in medical devices, with other activities that the European Union has been advancing in this area, showing that after marketing and commissioning, there is no additional regulated metrological control for most medical devices [3]. Consequently, the study highlighted the importance of generating models to estimate uncertainty in medical equipment and greater urgency in high use in clinical practice, such as sphygmomanometers. In this same sense, the National Institute of Metrology, Quality and Technology (INMETRO) of Brazil has developed regulations that delegate powers to a network of competent state institutes to carry out the mandatory verification of medical equipment used for weighing adults, pediatric scales, and sphygmomanometers [4]. It is reported that the lack of metrological control or verification, as well as the inadequate frequency of verification of these instruments, are generally the cause of measurement errors, followed by an erroneous or inadequate diagnosis, which puts the well-being of the patient at risk. In another work [5], it is reported that INMETRO uses a system to perform periodic verifications of measuring instruments, pointing to a legal metrological control where the model approval processes as recommended by OIML R16-1, show the absence of the estimate. uncertainty for the case of sphygmomanometers. Likewise, in Colombia there have been notable changes with respect to the standards aimed at technological management applicable to medical equipment, with a focus on legal metrological control, for which reason research on management models that integrate the requirements of the legal metrological control applied to biomedical equipment, and with the measurement processes involved in conformity assessment to support activities aimed at patient safety [6]. Other investigations [1] have focused on using statistical techniques to compare blood pressure measurements obtained with a radial sphygmomanometer with those obtained with two other types of non-invasive sphygmomanometers. In contrast, other studies [7] focus on the implications of errors in the calibration of sphygmomanometers in primary care. However, these investigations highlighting the importance of the measurement results do not direct their attention to the uncertainty estimation models for this type of equipment.
The International Organization of Legal Metrology (OIML) issued the international recommendation OIML R16-1 “Non-invasive mechanical sphygmomanometers” with specifications on general requirements, performance, efficiency, and mechanical and electrical safety, including test methods for type approval, applicable both to non-invasive mechanical sphygmomanometers, as well as their accessories that facilitate the non-invasive measurement of blood pressure using an inflatable cuff [8]. However, the metrological requirements section does not specify the estimation of uncertainty during the sphygmomanometer calibration process. In [9], a more effective approach for the legal metrological control of sphygmomanometers is presented, as established in OIML R-16: 2002, with a proposal to estimate uncertainty. However, not all sources are considered, which can affect the measurement in the calibration process.
This article presents a procedure based on the physical and mathematical model for estimating uncertainty, aiming to cover the need to know more precisely the measurement process of sphygmomanometers by proposing a structure with an approximation to the different sources that send the non-invasive blood pressure calibration process. We use the non-stochastic methodology called Guide to the Expression of Uncertainty in Measurement (GUM), and we take what is established in the international recommendation OIML R16-1 as a principle.
Materials and methods
In addition to exposing the materials and methods used to perform the calibration of the sphygmomanometer, we also include the method used to propose the estimation of uncertainty as established in the Guide to the Expression of Uncertainty in Measurement (GUM), which is based on the international recommendation OIML R16-1: 2002 [8].
Technical specifications
To perform the calibration, a generic description of the operation is used through direct comparison as a measurement method [10], obtained by observing directly in an instrument under test compared by a measurement instrument designed to measure magnitudes of the same nature as a reference [11]. For this calibration, the indication of the sphygmomanometer is used, compared with a reference manometer used as a standard, which, in metrological terms, has better technical specifications, which allows it to be selected as a reference standard, based on what is established in the OIML R- 16: 2002 [8]. To perform the data collection exercise and compare the results obtained, four (4) sphygmomanometers of different brands were randomly selected with previous calibrations every six months and have not been repaired.
Before starting the measurement process, and per the provisions of OIML R 16-1: 2002 [8], an air leak test of the pneumatic system must be performed. It is established that such a test must be carried out over the entire measurement range of the instrument under test, at at least five equidistant pressure points (for example, 7 kPa (50 mmHg), 13 kPa (100 mmHg), 20 kPa (150mmHg), 27kPa (200mmHg) and 34kPa (250mmHg)); allowing each value to stabilize. The leakage should not exceed 0.5 kPa / min (4 mmHg / min), if the instrument exceeds the maximum error allowed in the leak test, this should be recorded, and the equipment rejected. Additionally, an inspection of the zero must be carried out, where it is identified if the instrument has zero, as shown in figure 1. If so, the measurement of this is carried out. This measurement's maximum permissible error must not exceed ± 0.4 kPa (± 3 mmHg); otherwise, only visual inspection is carried out, and a record must be left.
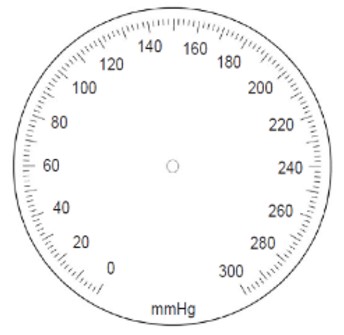 Figure 1.
Example of a manometer aneroid scale (divided in mmHg without a zero-tolerance zone)
Figure 1.
Example of a manometer aneroid scale (divided in mmHg without a zero-tolerance zone)
Source: OIML R 16-1, 2002
To perform the calibration through direct comparison and as established in the OIML R 16-1: 2002 recommendation [8], a calibrated reference manometer must be available, and the expanded uncertainty of the calibration certificate must be less than ± 0.1 kPa (± 0.8 mmHg), plus a pneumatic pump that generates a pressure of up to 4136 kPa (600 psi). The following reference manometer and pneumatic pump were used to take measurements in this research, as established in table 1.
Table 1.
Materials

Source: own work
Table 2.
Equipment under test
Source: own work
Complementary to the materials described in table 1 and table 2, some auxiliary materials, and tools such as connectors and conduction hoses are used to make the connections between the instruments; a stopwatch to perform the leak tests and the calibration process; and a rigid structure, to eliminate the difference in heights between the sphygmomanometer and the reference manometer.
Environmental conditions and permitted ranges
It is necessary to measure the temperature and relative humidity of the place where the calibration is carried out to confirm that this place does not exceed the temperature and humidity conditions described in this section. Therefore, it is necessary to have a traceable thermo-hygrometer that can measure environmental conditions during calibration. Additionally, the thermo-hygrometer must take the maximum value and the minimum value obtained during the process, and these data must be recorded. Similarly, the laboratory facilities' environmental conditions where the standard equipment is guarded must be taken, bearing in mind the permitted ranges as established in table 3.
Table 3.
Temperature and relative humidity

Source: OIML R 16-1, 2002
Consequently, the maximum allowed error criteria must be met at the time of the measurements, as established in table 4.
Table 4.
Ranges for maximum permissible errors
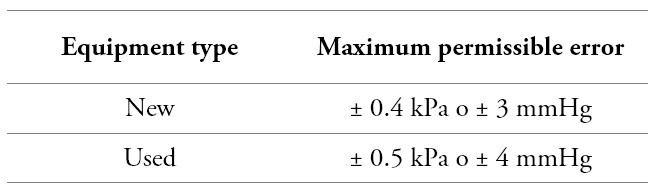
Source: OIML R 16-1, 2002
t is also important to consider the work reported in [9], in which the OIML R 16-1 is analyzed in detail. It is proposed to be established as a criterion for evaluating the conformity of equipment the imposition of a safety margin less than or equal to 30% of the tolerance interval [12], which for the legal metrological control applied to sphygmomanometers would correspond to 2.7 mmHg, as this value is associated with 30% of the difference between the maximum and minimum values of the diastolic pressure of the prehypertension.
Calibration method
The sphygmomanometer and the reference manometer are fixed in the rigid structure as shown in figure 2, the above to eliminate the uncertainty due to the difference in heights.
 Figure 2.
Sphygmomanometer calibration setup
Figure 2.
Sphygmomanometer calibration setup
Source: own work
The thermal balance between the sphygmomanometer, the reference manometer, and the environment must be monitored for 20 minutes until the environmental conditions stabilize in the intervals described in table 3. If it does not stabilize, the calibration cannot be performed. If these conditions are exceeded during the calibration, the calibration must be stopped and wait for the conditions to stabilize. When this happens, the calibration is started from zero. If, on the contrary, the conditions do not stabilize, it must be suspended. Subsequently, pre-charges must be performed to excite the system of the equipment under test at 50% and 90% or 100% of the scale, in addition to performing an air leak test as previously described in the technical specifications to do so, which should set the value to be measured on the standard instrument and count the time of the leak with the stopwatch. The leakage should not exceed 0.5 kPa / min (4 mmHg / min); If the instrument under test exceeds the maximum permissible error in the leak test, this should be recorded and rejected.
The measurement points are selected in intervals no greater than 7 kPa (50 mmHg), between 0 kPa (0 mmHg) and the maximum value of the sphygmomanometer scale, where the value that will be measured in the instrument underneath must first be set and then read the data on the reference manometer. After that, the first series’ data are taken in ascending order, and the observed values are recorded. To start the downward measurement of the first series, wait 1 minute at the maximum point of the equipment and record the data. Next, the next descending point is taken and so on until series 1 is completed. It takes 1 minute at the end of the data collection of series 1 and we begin again with the second series in an ascending manner [13].
Physical and mathematical model for estimating uncertainty
The physical measurement model consists of assumptions about the measurand itself and the relevant physical and chemical variables for the measurement. However simple it may be, a physical measurement has a model associated with it that only approximates the real process [14]. In this article, the physical and mathematical model was based on the phenomenon generated by the dynamic pressure in a sphygmomanometer, represented in the following equation.
Where
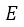 : Absolute error of the instrument under the test
: Absolute error of the instrument under the test
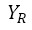 : Reference value
: Reference value
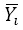 : Average of the instruments readings
: Average of the instruments readings
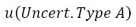 : Standard deviation of the data, divided by the total number of measurements taken
: Standard deviation of the data, divided by the total number of measurements taken
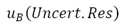 : Resolution of the instrument under test
: Resolution of the instrument under test
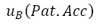 : Accuracy of the standard equipment
: Accuracy of the standard equipment
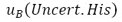 : Hysteresis of the instrument under test
: Hysteresis of the instrument under test
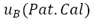 : Value extracted from the calibration certificate of the standard instrument
: Value extracted from the calibration certificate of the standard instrument
For the mathematical calculation of uncertainty, as specified by the GUM, it must be categorized as random sources (type A uncertainty) and systematic (type B uncertainty). The purpose of the classification in types A and B is to indicate two different ways of evaluating the uncertainty components. Type A components are expressed by estimated variances 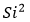 (or the estimated “standard deviations”
(or the estimated “standard deviations” 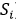 ), and the number of degrees of freedom
), and the number of degrees of freedom 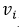 , where necessary, will be given the covariances. The components of type B must be expressed through estimated variances
, where necessary, will be given the covariances. The components of type B must be expressed through estimated variances 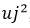 , which can be considered approximations to the corresponding variances, whose existence is assumed. The quantities
, which can be considered approximations to the corresponding variances, whose existence is assumed. The quantities 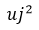 can be treated as variances and the as standard deviations. When necessary, covariances should be treated similarly [15]. In figure 3, the different sources in which the proposal for the estimation of the expanded uncertainty are composed are exposed, bearing in mind the physical phenomenon for sphygmomanometers’ calibration.
can be treated as variances and the as standard deviations. When necessary, covariances should be treated similarly [15]. In figure 3, the different sources in which the proposal for the estimation of the expanded uncertainty are composed are exposed, bearing in mind the physical phenomenon for sphygmomanometers’ calibration.
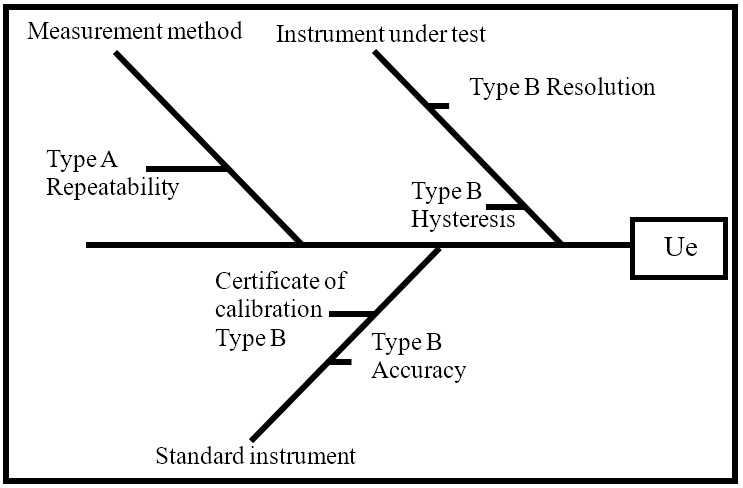 Figure 3.
Sources of uncertainty calibration sphygmomanometer
Figure 3.
Sources of uncertainty calibration sphygmomanometer
Source: own work
Considering the above, equations (2) and (3) represent the determination of the type A uncertainty by the repeatability of the measurements.
Where
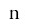 : Total number of measurements taken.
: Total number of measurements taken.
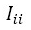 : It is each of the registered standard readings.
: It is each of the registered standard readings.
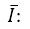 : Average of the readings.
: Average of the readings.
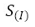 : Standard deviation of the data.
: Standard deviation of the data.
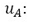 : Type A uncertainty, due to data variation.
: Type A uncertainty, due to data variation.
We analyzed how the different instruments and the physical phenomenon intervene in the measurement system to determine the various type B uncertainty sources, ascertaining the systematic components presented in table 5.
Table 5.
Components of Type B uncertainty

Source: own work
Consequently, the sensitivity coefficients, which describe how sensitive the measurand is regarding the corresponding input magnitude variations, must be calculated. In other words, it is expressed as the output estimate and varies with changes in the input estimates 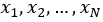 [15]. As there is a functional relationship through a mathematical model for the measurand
[15]. As there is a functional relationship through a mathematical model for the measurand 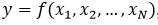 , for this investigation, the sensitivity coefficient
, for this investigation, the sensitivity coefficient 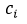 can be estimated by the partial derivative of
can be estimated by the partial derivative of 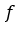 regarding
regarding 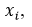
And according to the law of propagation of uncertainties, the expression for the combined standard uncertainty 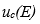 (it is assumed that there is no correlation between the variables) is:
(it is assumed that there is no correlation between the variables) is:
When applying equations (8) and (9) to the sphygmomanometer calibration process and considering both the sources of uncertainty and the mathematical model expressed in equation (1), we obtain equation (10).
The estimate of the effective degrees of freedom of the standard uncertainty 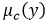 associated with the output estimate is obtained using the Welch-Satterthwaite formula.
associated with the output estimate is obtained using the Welch-Satterthwaite formula.
After applying equation (12), once the effective value 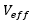 has been obtained, this value must be compared in the Student t distribution table to obtain the coverage factor (k) evaluated for a coverage probability of 95.45%, which must be multiplied by the combined uncertainty
has been obtained, this value must be compared in the Student t distribution table to obtain the coverage factor (k) evaluated for a coverage probability of 95.45%, which must be multiplied by the combined uncertainty 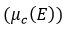 to estimate the result of the expanded uncertainty with equation (13).
to estimate the result of the expanded uncertainty with equation (13).
Where
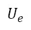 : Expanded uncertainty
: Expanded uncertainty
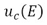 : Combined uncertainty
: Combined uncertainty
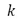 : Coverage factor
: Coverage factor
Finally, one of the results obtained in this research was the proposed methodology for estimating the uncertainty in the calibration of sphygmomanometers under OIML R16-1 from a legal metrology perspective using the GUM method, as presented in figure 4.
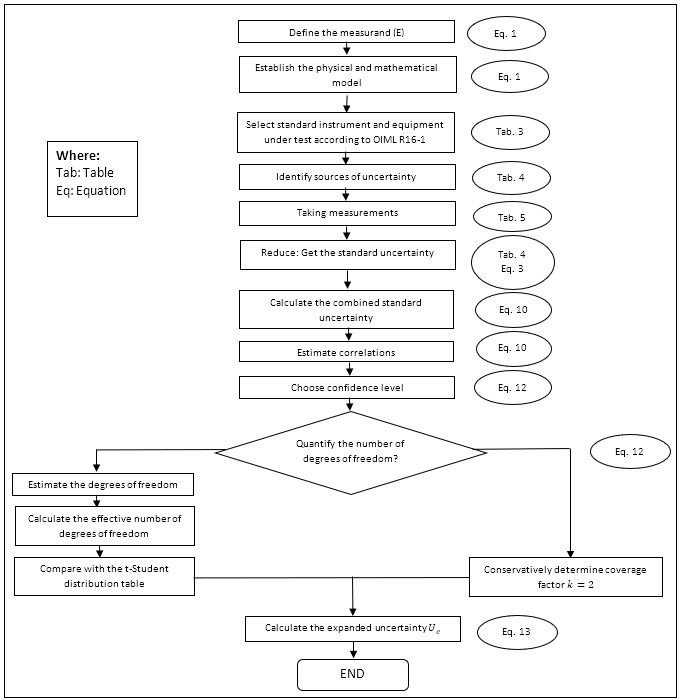 Figure 4.
Schematic diagram for estimating the uncertainty using the GUM method
Figure 4.
Schematic diagram for estimating the uncertainty using the GUM method
Source: own work
Results
The estimation of uncertainty in the calibration is proposed according to what is established in a non-stochastic methodology such as the Guide to the Expression of Uncertainty in Measurement (GUM) [15]. This has been used in different processes from analytical chemistry tests [16], gamma-ray spectrometry [17], as well as in the application in measuring instruments such as electromagnetic compatibility tests [18].
Estimation of uncertainty in sphygmomanometers
Measurements were taken as proposed in the method presented above, and the results are presented in the following tables. For each of the exercises, two series of ascending and descending sequences were performed. Finally, four (4) measurements were obtained, and the measurement error was calculated for each of the points. The measurement method and the points selected to carry out the calibration process were taken according to the laboratory experience, bearing in mind that the points encompass the physiological behavior of blood pressure since it includes points from healthy, hypertensive, or hypotensive patients. The number of points was considered according to the guidelines defined in DKD-R 6-1 [13], where it is established to perform the calibration in two sequences for each point (2 ascending and 2 descending) and at the maximum point of the equipment.
Table 6.
Measurement data of a sphygmomanometer 1
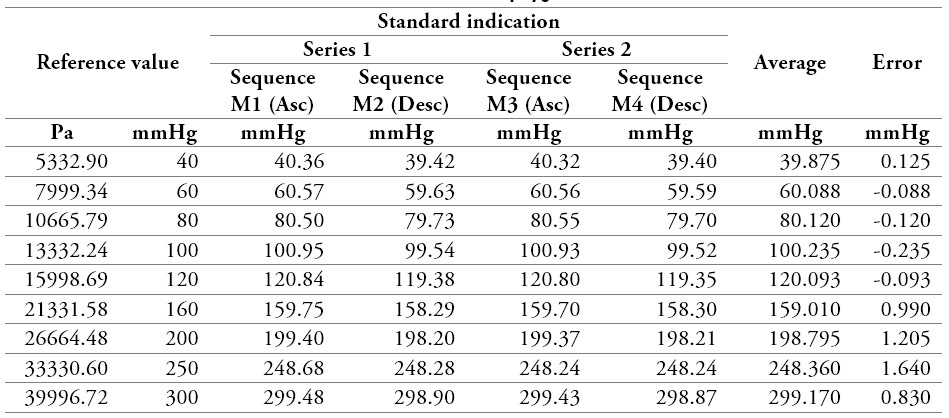
Source: own work
Table 7.
Measurement data of a sphygmomanometer 2
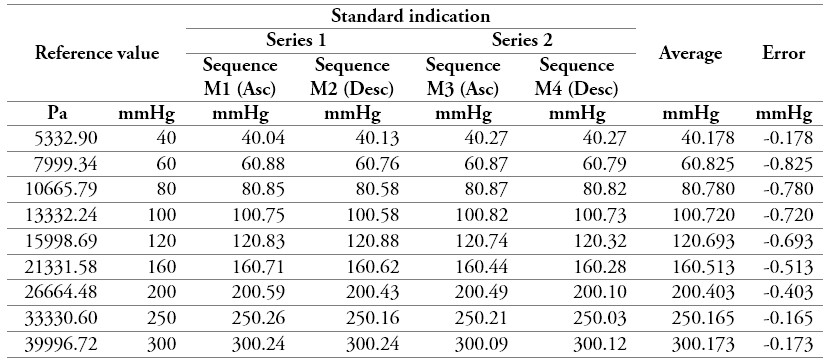
Source: own work
Table 8.
Measurement data of a sphygmomanometer 3
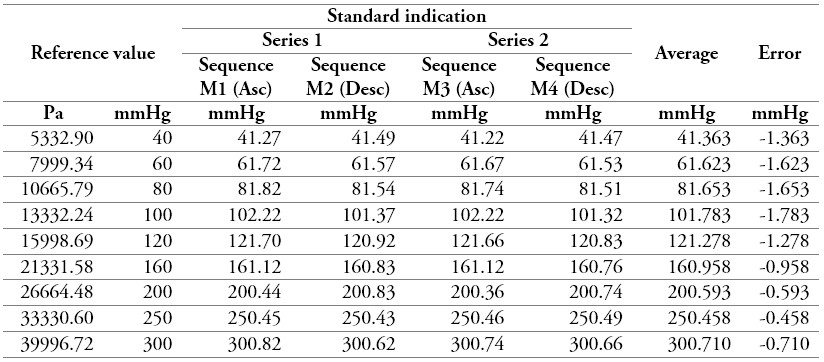
Source: own work
Table 9.
Measurement data of a sphygmomanometer 4
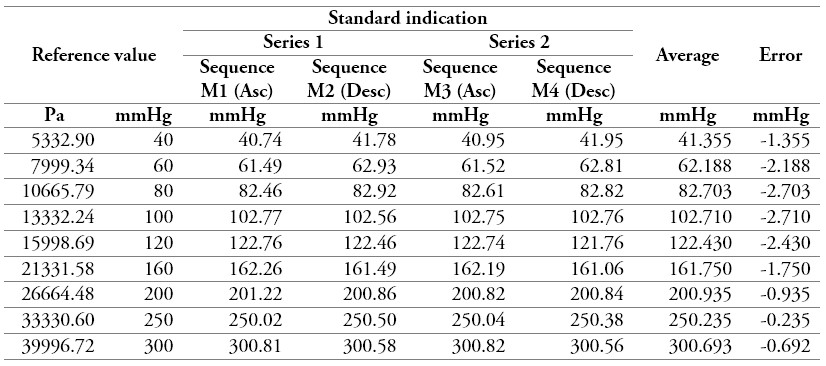
Source: own work
After obtaining the results in the measurement process, the uncertainty in the sphygmomanometer calibration process is estimated by applying equations (3), (4), (5), (6), (7), (10), (12), and (13) to the data in table 6, table 7, table 8, and table 9. The estimated expanded uncertainty 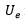 for each measurement point is from 40 mmHg to 300 mmHg, expressed in the following tables.
for each measurement point is from 40 mmHg to 300 mmHg, expressed in the following tables.
Table 10.
Results of the calibration of a sphygmomanometer 1
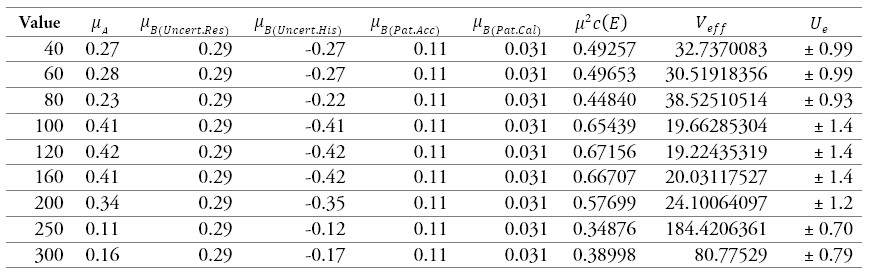
Source: own work
Table 11.
Results of the calibration of a sphygmomanometer 2
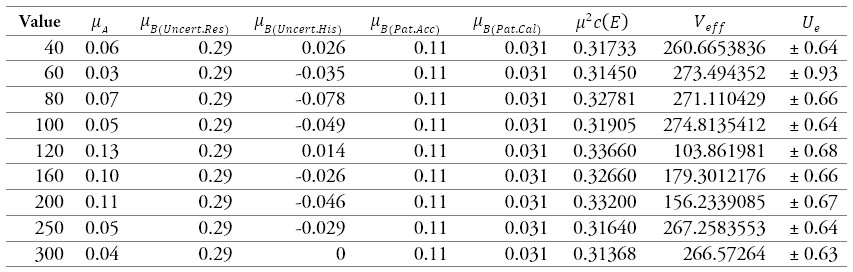
Source: own work
Table 12.
Results of the calibration of a sphygmomanometer 3
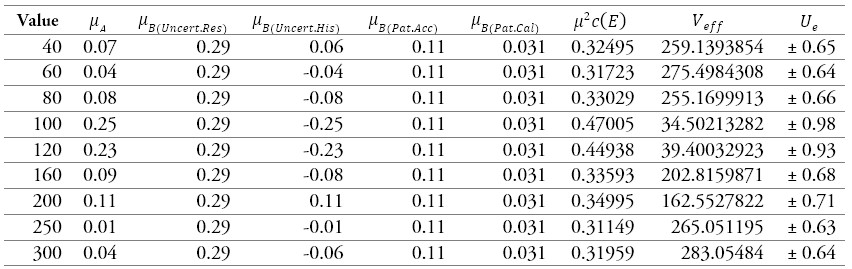
Source: own work
Table 13.
Results of the calibration of a sphygmomanometer 4
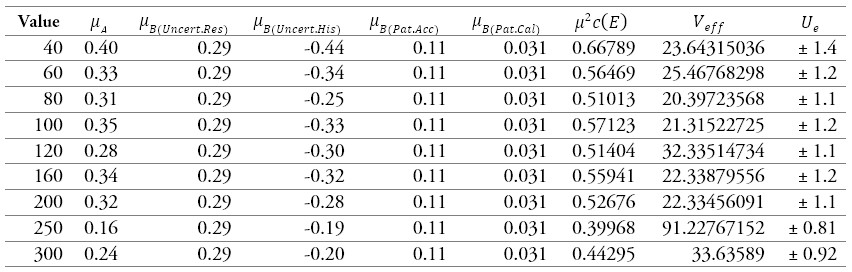
Source: own work
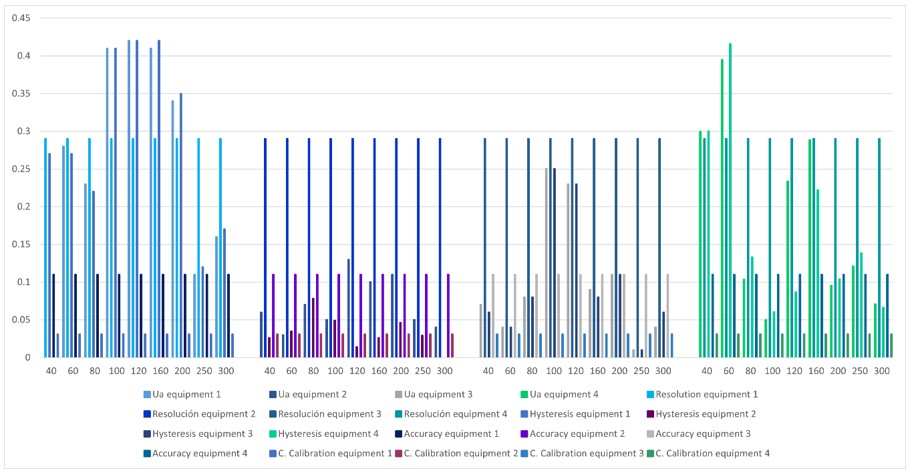 Figure 5.
Contribution of uncertainty for each of the selected equipment
Figure 5.
Contribution of uncertainty for each of the selected equipment
Source: own work
Each of the graphs with their respective analysis are detailed below.
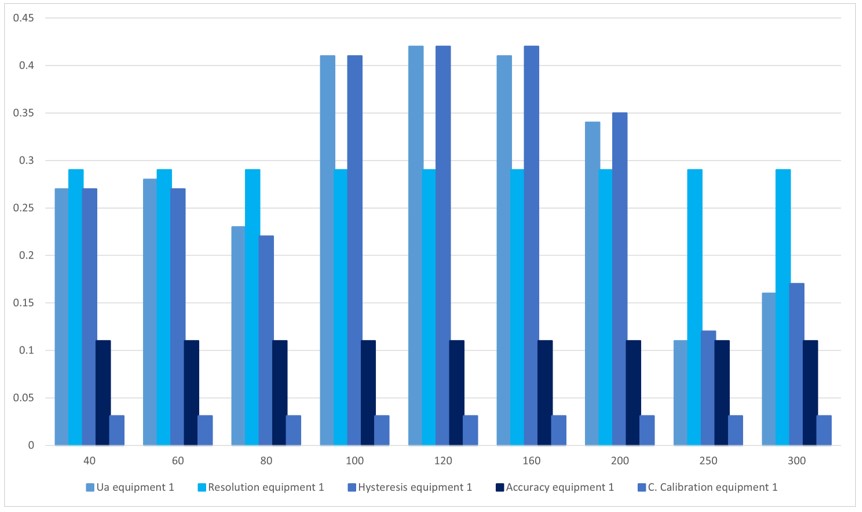 Figure 6.
Contribution of uncertainty, equipment 1
Figure 6.
Contribution of uncertainty, equipment 1
Source: own work
Table 10 and figure 6 are the sources of uncertainties that most contribute to the process. The uncertainty due to the sphygmomanometer's resolution predominates at 40 mmHg, 60 80 mmHg, 250 mmHg, and 300 mmHg. For the points 100 mmHg and 120 mmHg, the uncertainty that contributes the most is data repeatability and hysteresis for 160 mmHg and 200 mmHg. The uncertainty that contributes the most is due to the hysteresis of the sphygmomanometer.
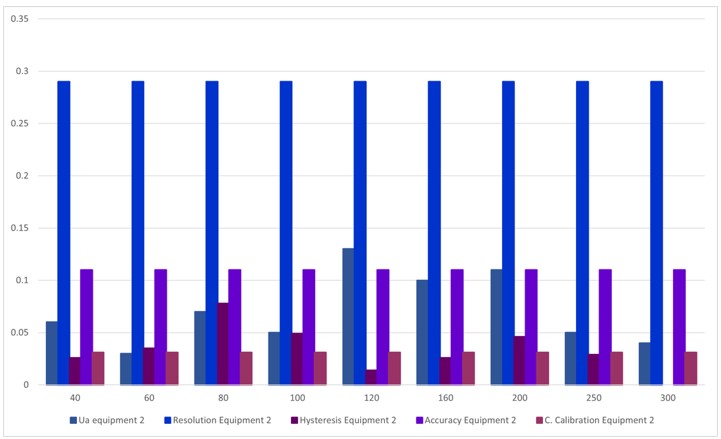 Figure 7.
Contribution of uncertainty, Equipment 2
Figure 7.
Contribution of uncertainty, Equipment 2
Source: own work
In table 11 and figure 7, the sources of uncertainties that contribute the most to the process, where the uncertainty due to the sphygmomanometer's resolution predominates at all points.
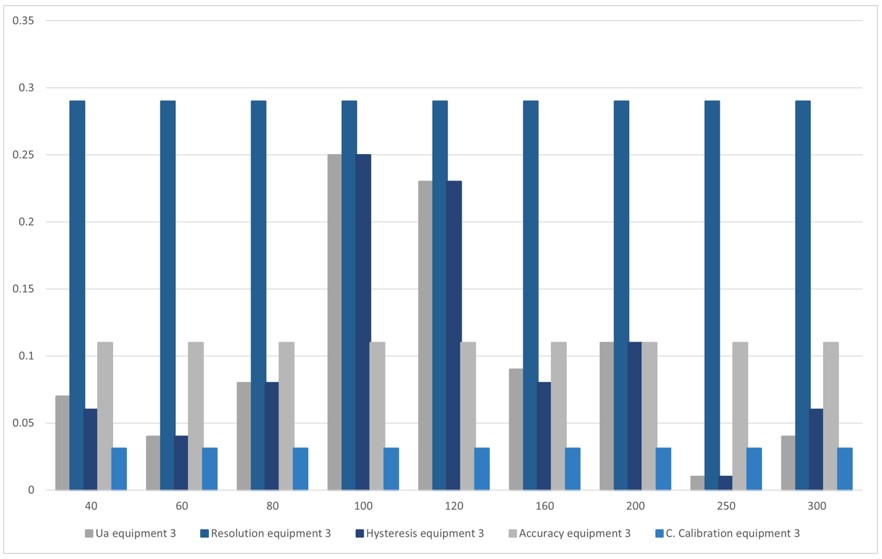 Figure 8.
Contribution of uncertainty, Team 3
Figure 8.
Contribution of uncertainty, Team 3
Source: own work
Table 12 and figure 8 show the sources of uncertainties that contribute the most to the process, where the uncertainty due to the resolution of the sphygmomanometer prevails at all points.
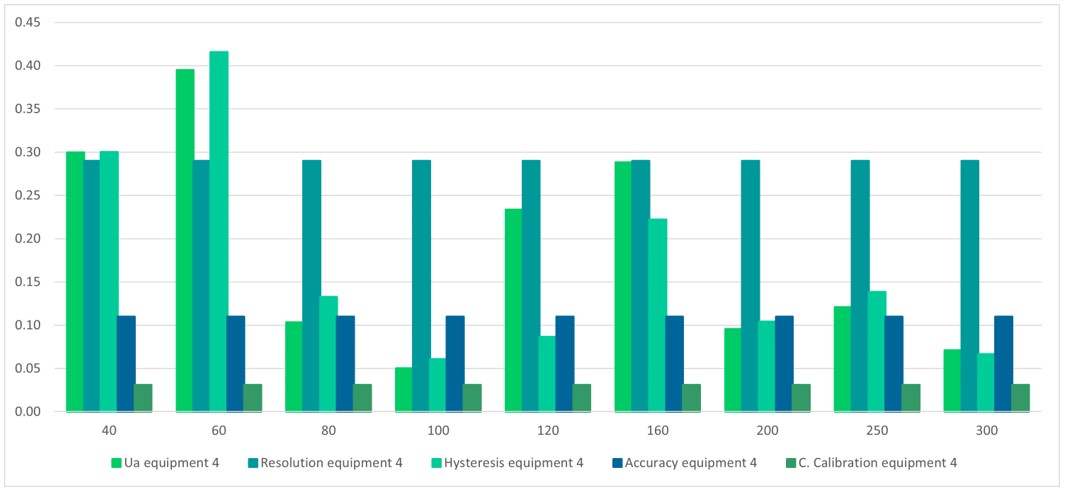 Figure 9.
Contribution of uncertainty, equipment 4
Figure 9.
Contribution of uncertainty, equipment 4
Source: own work
Table 13 and figure 9 show the sources of uncertainties that contribute the most to the process, where the uncertainty due to the resolution of the sphygmomanometer predominates at the points 80 mmHg, 100 mmHg, 120 mmHg, 200 mmHg, 250 mmHg, and 300 mmHg, which correspond to most of the points. For the points 40 mmHg and 60 mmHg, and 160 mmHg, the uncertainty that contributes the most is data repeatability and hysteresis.
Discussion
According to the results obtained in each of the calibrations carried out, similar behavior is observed in the different instruments under test, in which the sources determined in the mathematical model highlight the different components. This allows us to get closer to the real behavior of the equipment and an assurance of its measurements.
Thus, in figure 10, it can be observed that for equipment 1, the predominant uncertainty in most of the points is the equipment's resolution, followed by repeatability and hysteresis, while for equipment 2 and 3, the uncertainty prevails due to the team resolution at all points. According to the above, it can be inferred that equipment 1 had a greater variation in the data obtained compared to 2 and 3 as they had greater stability in the behavior of the data since the repeatability and hysteresis uncertainty is lower at most points. Comparing the 3, it is observed that the team with the greatest stability in the data is equipment 2 since in equipment 3, the uncertainty due to repeatability and hysteresis is greater at the points 100 mmHg and 120 mmHg than at the rest of the points.
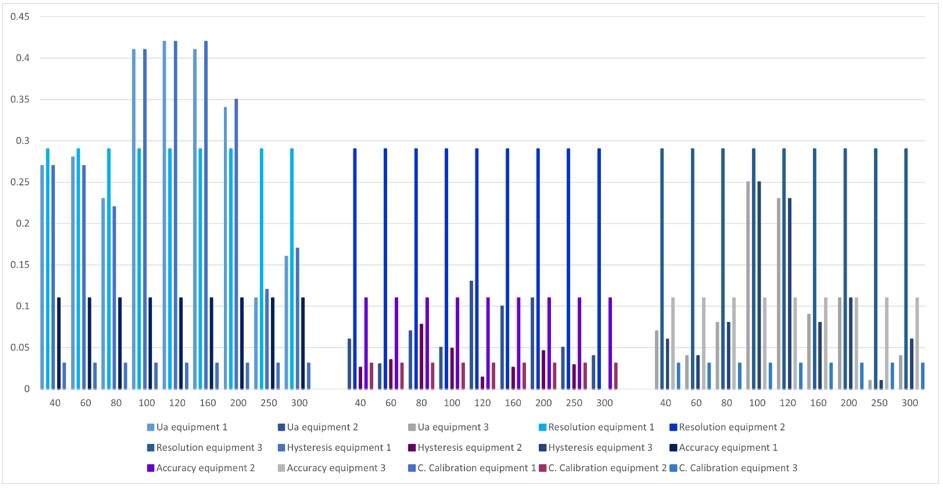 Figure 10.
Comparison between equipment 1, 2 and 3
Figure 10.
Comparison between equipment 1, 2 and 3
Source: own work
In figure 11, equipment 1, compared to equipment 4, presents a greater variation in the behavior of the data, since only in the last two points it does not present a high uncertainty due to repeatability and hysteresis. For equipment 4, it only occurs in the initial points and the two intermediate points. When comparing with the rest of equipment 4, it is observed that the uncertainty by team resolution predominates.
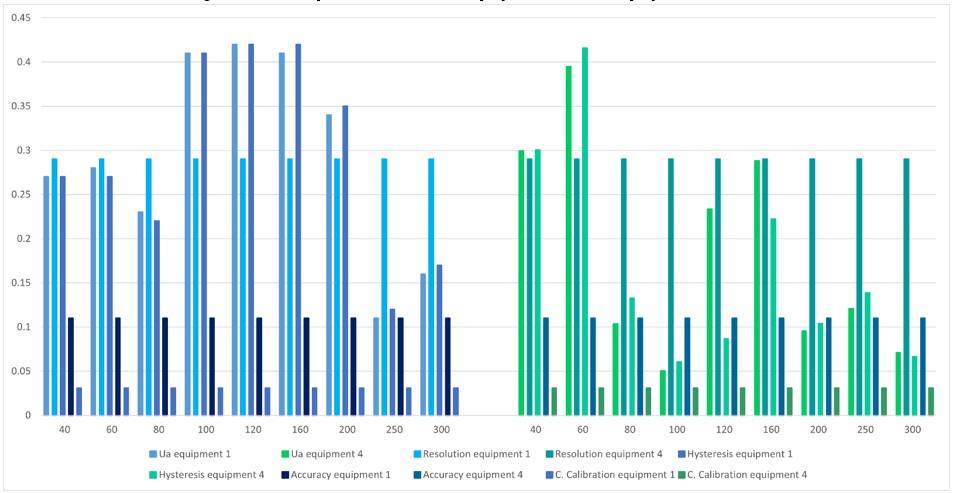 Figure 11.
Comparison between equipment 1 and equipment 4
Figure 11.
Comparison between equipment 1 and equipment 4
Source: own work
In figure 12, equipment 2 and 3 have a more stable behavior than equipment 4. The above is because the uncertainty by repeatability and hysteresis is lower in most of the points. For equipment 3, it is observed that at the points 100 mmHg and 120 mmHg, the uncertainty due to repeatability and hysteresis is greater than in the rest of the points.
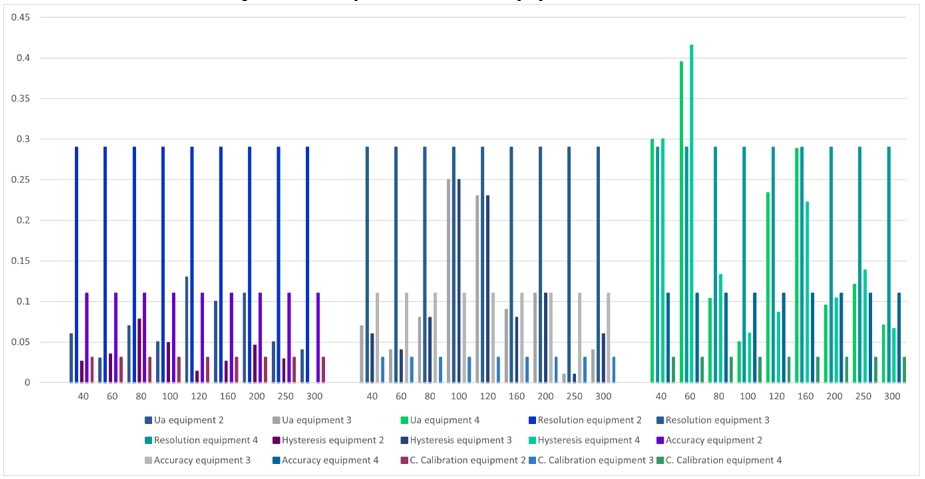 Figure 12.
Comparison between equipment 2, 3 and 4
Figure 12.
Comparison between equipment 2, 3 and 4
Source: own work
According to the discussion carried out previously, the predominant uncertainty in most cases is by the resolution of the equipment, followed by the behavior of the equipment in the repeatability and hysteresis test.
Conclusions
This article estimates the uncertainty in the calibration of sphygmomanometers under OIML R16-1 from a legal metrology perspective. Identifying sources of uncertainty has not been perceived or analyzed in most of the research studies reported in the literature. The experimental results show that derived from the equipment specifications under test, the uncertainty that contributes the most to each of the calibrations is the resolution of the sphygmomanometer followed by the uncertainty due to repeatability or hysteresis, which is associated with the behavior of the equipment. The novelty factor of the proposed methodology consists in showing the advantage of estimating uncertainty through a structure that combines the mathematical model under a non-stochastic method according to the GUM with the international standard OIML R16 (which has gone through different reviews), which provides consistent execution activities aligned with the regulatory standard to be performed in the calibration of sphygmomanometers, with the additional possibility of expanding the scope to processes related to model approval. Likewise, the importance of ensuring the results in medical equipment, specifically in sphygmomanometers, within the framework of model approval processes and the impact that these activities can have on patient safety is highlighted.
The consistent results in the different equipment under test carried out allow its application to deliver pertinent information to the healthcare part for decision-making when calibrating sphygmomanometers as diagnostic equipment. Future work remains to carry out a multivariate analysis with different calibration methodologies that define the correlation between the different sources of uncertainty and statistically determine the values of errors and expanded uncertainty, independent of brands and models of sphygmomanometers.
References
[1] H. H. Rachmat, “Comparison of radialis sphygmomanometer in evaluating the blood pressure of healthy volunteers,” in 1st International Conference on Biomedical Engineering (IBIOMED), Yogyakarta, Indonesia, 2016, pp. 1-4. https://doi.org/10.1109/IBIOMED.2016.7869817
[2] A. Badnjevic, L. Gurbeta-Pokvic, D. Boskovic, and Z. Dzemic, “Medical devices in legal metrology,” de 4th Mediterranean Conference on Embedded Computing MECO 2015, Budva, Montenegro, 2015, pp. 365-367. https://doi.org/10.1109/MECO.2015.7181945
[3] M. do Céu Ferreira, “The role of metrology in the field of medical devices,” Int. J. Metrol. Qual. Eng., vol. 2, no. 2, pp. 135-140, 2011. https://doi.org/10.1051/ijmqe/2011101
[4] M. R. Paiva, O. Pohlmann-Filho, and A. Soratto, “Prospection for metrological control in medical scales and sphygmomanometers in the state of Santa Catarina-Brazil,” J. Phys.: Conf. Ser., vol. 575, no. 1, pp. 24-27, 2013. https://doi.org/10.1088/1742-6596/575/1/012047
[5] A. Soratto, O. Pohlmann-Filho, M. R. Paiva, R. B. Giordani, and C. Bringhenti, “Development of a system to increase the legal metrological control of measuring instruments in Brazil,” Int. J. Metrol. Qual. Eng., vol. 5, no. 3, p. 304, 2014. https://doi.org/10.1051/ijmqe/2014013
[6] A. F. Ramírez-Barrera, J. F. Martínez-Gómez, and E. Hidalgo-Vásquez, “Management Model for the Application of Legal Metrological Control and Conformity Assessment in Biomedical Equipment,” Revista Ingeniería Biomédica, vol. 11, no. 21, pp. 73-80, 2017. https://doi.org/10.24050/19099762.n21.2017.1175
[7] A. Rouse and T. Marshall, “The extent and implications of sphygmomanometer calibration error in primary care,” J. Hum. Hypertens., vol. 15, no. 9, pp. 587-591, 2001. https://doi.org/10.1038/sj.jhh.1001241
[8] International Organization of Legal Metrology, OIML R 16-1: Non-invasive mechanical sphygmomanometers, 2002. https://www.oiml.org/en/files/pdf_r/r016-1-e02.pdf/at_download/file
[9] C. H. d. M. Fraga, and R. F. Farias, “A more effective approach to the legal metrological control of sphygmomanometer,” OIML Bulletin, vol. 58, no. 4, pp. 5-9, 2017. https://www.oiml.org/en/publications/bulletin/pdf/oiml_bulletin_october_2017.pdf
[10] A. Giordani and L. Mari, “A structural model of direct measurement,” Measurement, vol. 145, pp. 535-550, 2019. https://doi.org/10.1016/j.measurement.2019.05.060
[11] Joint Committee for Guides in Metrology, JCGM 200: International vocabulary of metrology – Basic and general concepts and associated terms (VIM), 2012. https://www.bipm.org/utils/common/documents/jcgm/JCGM_200_2012.pdf
[12] Joint Committee for Guides in Metrology, JCGM 106: Evaluation of measurement data – The role of measurement uncertainty in conformity assessment, 2012. https://www.bipm.org/utils/common/documents/jcgm/JCGM_106_2012_E.pdf
[13] PhysikalischTechnische Bundesanstalt, DKD-R 6-1: Calibration of Pressure Gauges, 2014.
[14] L. Kirkup and R. B. Frenkel, An introduction to uncertainty in measurement using the gum (Guide to the Expression of Uncertainty in Measurement), Cambridge University Press, 2006. https://doi.org/10.1017/CBO9780511755538
[15] Joint Committee for Guides in Metrology, JCGM 100: Evaluation of measurement data – Guide to the expression of uncertainty in measurement, 2008. https://ncc.nesdis.noaa.gov/documents/documentation/JCGM_100_2008_E.pdf
[16] J. Sousa, A. M. Reynolds, and Á. S. Ribeiro, “A comparison in the evaluation of measurement uncertainty in analytical chemistry testing between the use of quality control data and a regression analysis,” Accred. Qual. Assur., vol. 17, no. 2, pp. 207-214, 2012. https://doi.org/10.1007/s00769-011-0874-y
[17] O. Sima and M. C. Lépy, “Application of GUM Supplement 1 to uncertainty of Monte Carlo computed efficiency in gamma-ray spectrometry,” Applied Radiation and Isotopes, vol. 109, pp. 493-499, 2016. https://doi.org/10.1016/j.apradiso.2015.11.097
[18] M. Azpurua, C. Tremola, and E. J. Paez-Barrios, “Comparison of the GUM and Monte Carlo Methods for the Uncertainty Estimation in Electromagnetic Compatibility Testing,” Progress in Electromagnetics Research B, vol. 34, pp. 125-144, 2011. https://doi.org/10.2528/PIERB11081804
Notes
*
This article was carried out within the framework of a research project called “Normative harmonization of legal metrology through a management model applied to the health sector in Colombia” with code P17202, funded by the Instituto Tecnológico Metropolitano ITM (Medellín-Colombia), and the data obtained were carried out in the ITM Metrology Laboratory, in the General Hospital of Medellín.
Author notes
a Corresponding author. E-mail: andresramirez@itm.edu.co
Additional information
How to cite this article: A. F. Ramírez-Barrera, E. Delgado-Trejos, and V. Ramírez-Gómez, “Uncertainty estimation in the sphygmomanometers calibration according to OIML R16-1 from a legal metrology perspective,” Ing. Univ., vol. 25, 2021 [Online]. https://doi.org/10.11144/Javeriana.iued25.uesc









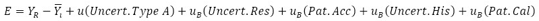 (1)
(1)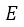 : Absolute error of the instrument under the test
: Absolute error of the instrument under the test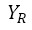 : Reference value
: Reference value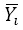 : Average of the instruments readings
: Average of the instruments readings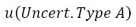 : Standard deviation of the data, divided by the total number of measurements taken
: Standard deviation of the data, divided by the total number of measurements taken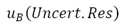 : Resolution of the instrument under test
: Resolution of the instrument under test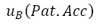 : Accuracy of the standard equipment
: Accuracy of the standard equipment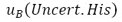 : Hysteresis of the instrument under test
: Hysteresis of the instrument under test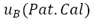 : Value extracted from the calibration certificate of the standard instrument
: Value extracted from the calibration certificate of the standard instrument 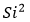 (or the estimated “standard deviations”
(or the estimated “standard deviations” 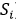 ), and the number of degrees of freedom
), and the number of degrees of freedom 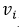 , where necessary, will be given the covariances. The components of type B must be expressed through estimated variances
, where necessary, will be given the covariances. The components of type B must be expressed through estimated variances 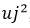 , which can be considered approximations to the corresponding variances, whose existence is assumed. The quantities
, which can be considered approximations to the corresponding variances, whose existence is assumed. The quantities 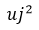 can be treated as variances and the as standard deviations. When necessary, covariances should be treated similarly [
can be treated as variances and the as standard deviations. When necessary, covariances should be treated similarly [
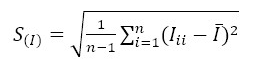 (2)
(2)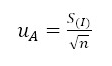 (3)
(3)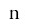 : Total number of measurements taken.
: Total number of measurements taken.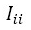 : It is each of the registered standard readings.
: It is each of the registered standard readings. 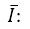 : Average of the readings.
: Average of the readings.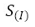 : Standard deviation of the data.
: Standard deviation of the data.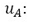 : Type A uncertainty, due to data variation.
: Type A uncertainty, due to data variation.
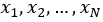 [
[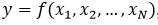 , for this investigation, the sensitivity coefficient
, for this investigation, the sensitivity coefficient 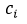 can be estimated by the partial derivative of
can be estimated by the partial derivative of 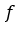 regarding
regarding 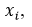
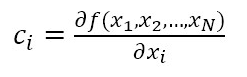 (8)
(8) (it is assumed that there is no correlation between the variables) is:
(it is assumed that there is no correlation between the variables) is: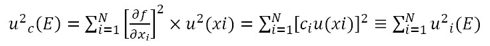 (9)
(9)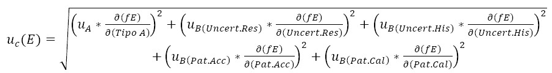 (10)
(10)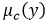 associated with the output estimate is obtained using the Welch-Satterthwaite formula.
associated with the output estimate is obtained using the Welch-Satterthwaite formula.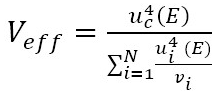 (11)
(11)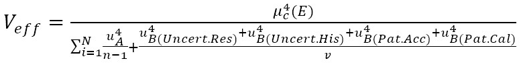 (12)
(12)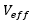 has been obtained, this value must be compared in the Student t distribution table to obtain the coverage factor (k) evaluated for a coverage probability of 95.45%, which must be multiplied by the combined uncertainty
has been obtained, this value must be compared in the Student t distribution table to obtain the coverage factor (k) evaluated for a coverage probability of 95.45%, which must be multiplied by the combined uncertainty 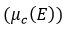 to estimate the result of the expanded uncertainty with equation (
to estimate the result of the expanded uncertainty with equation (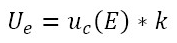 (13)
(13)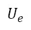 : Expanded uncertainty
: Expanded uncertainty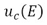 : Combined uncertainty
: Combined uncertainty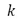 : Coverage factor
: Coverage factor




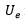 for each measurement point is from 40 mmHg to 300 mmHg, expressed in the following tables.
for each measurement point is from 40 mmHg to 300 mmHg, expressed in the following tables.











