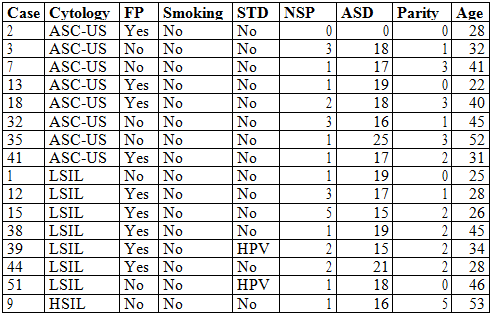
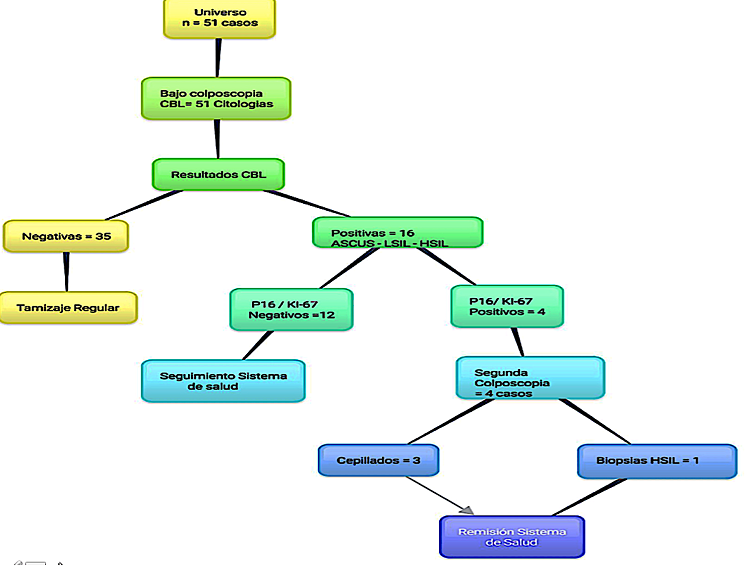
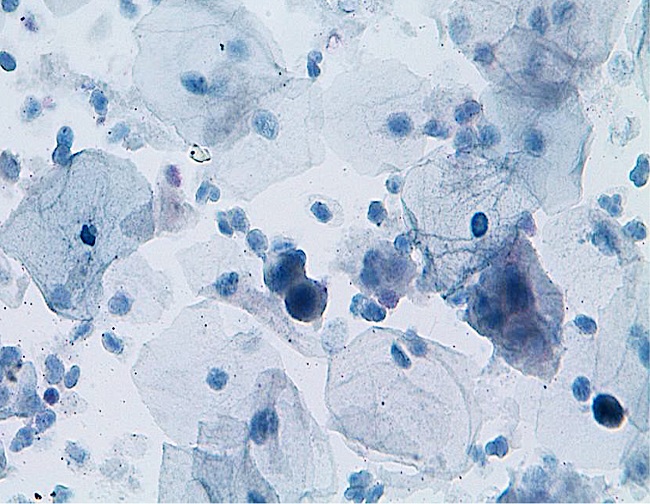
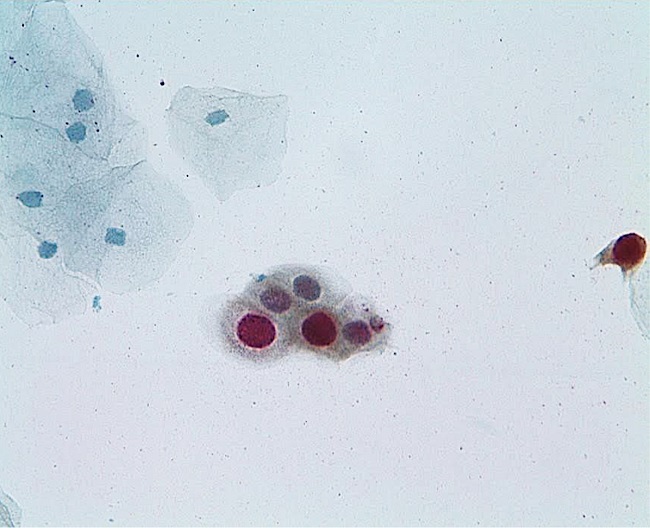
To the participating
patients. To Dr.
Juan Carlos Bonilla for reading the samples as a pathologist.
1.Bosch FX, Manos MM, Muñoz N, Sherman M, Jansen AM, Peto J, et al. Prevalence of human papillomavirus in cervical cancer: a worldwide perspective. International biological study on cervical cancer (IBSCC) Study Group. J Natl Cancer Inst. 1995;87(11):796-802.
2.Walboomers JM, Jacobs MV, Manos MM, Bosch FX, Kummer JA, Shah KV, et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol. 1999;189(1):12-9.
3.Wright JD, Rader JS, Dávila R, Powell MA, Mutch DG, Gao F, et al. Human papillomavirus triage for young women with atypical squamous cells of undetermined significance. Obstet Gynecol. 2006;107(4):822-9.
4.Tornesello ML, Buonaguro L, Giorgi-Rossi P, Buonaguro FM. Viral and cellular biomarkers in the diagnosis of cervical intraepithelial neoplasia and cancer. BioMed research international. 2013;2013:519619. https://doi.org/10.1155/2013/519619.
5.Li N, Franceschi S, Howell-Jones R, Snijders PJ, Clifford GM. Human papillomavirus type distribution in 30,848 invasive cervical cancers worldwide: Variation by geographical region, histological type and year of publication. Int J Cancer. 2011;128(4):927. https://doi.org/10.1002/ijc.25396.
6.Hopman AH, Smedts F, Dignef W, Ummelen M, Sonke G, Mravunac M, et al. Transition of high-grade cervical intraepithelial neoplasia to micro-invasive carcinoma is characterized by integration of HPV 16/18 and numerical chromosome abnormalities. J Pathol. 2004;202(1):23-Epub 2003/12/25.
7.Dürst M, Dzarlieva-Petrusevska RT, Boukamp P, Fusenig NE, Gissmann L. Molecular and cytogenetic analysis of immortalized human primary keratinocytes obtained after transfection with human papillomavirus type 16 DNA. Oncogene. 1987;1(3):251-6.
8.Werness BA, Levine AJ, Howley PM. Association of human papillomavirus types 16 and 18 E6 proteins with p53. Science. 1990;248(4951):76-9.
9.Dyson N, Howley PM, Münger K, Harlow E. The human papilloma virus-16 E7 oncoprotein is able to bind to the retinoblastoma gene product. Science. 1989;243(4893):934-7.
10.Toro de Méndez M, Ferrández Izquierdo A. [Detection of human papilloma virus (HPV) in liquid-based cervical samples. Correlation with protein p16INK4a expression]. Invest Clin. 2011;52(1):3-14.
11.Nakao Y, Yang X, Yokoyama M, Ferenczy A, Tang SC, Pater MM, et al. Induction of p16 during immortalization by HPV 16 and 18 and not during malignant transformation. British journal of cancer. 1997;75(10):1410-Epub 1997/01/01.
12.Shidham VB, Mehrotra R, Varsegi G, D'Amore KL, Hunt B, Narayan R. p16 immunocytochemistry on cell blocks as an adjunct to cervical cytology: Potential reflex testing on specially prepared cell blocks from residual liquid-based cytology specimens. Cytojournal. 2011;8. https://doi.org/10.4103/1742-6413.76379
13.Singh M, Mockler D, Akalin A, Burke S, Shroyer A, Shroyer KR. Immunocytochemical colocalization of P16(INK4a) and Ki-67 predicts CIN2/3 and AIS/adenocarcinoma. Cancer cytopathology. 2012;120(1):26. https://doi.org/10.1002/cncy.20188
14.Donà MG, Vocaturo A, Giuliani M, Ronchetti L, Rollo F, Pescarmona E, et al. p16/Ki-67 dual staining in cervico-vaginal cytology: correlation with histology, Human Papillomavirus detection and genotyping in women undergoing colposcopy. Gynecol Oncol. 2012;126(2):198-202. https://doi.org/10.1016/j.ygyno.2012.05.004
15.Bergeron C, Ordi J, Schmidt D, Trunk MJ, Keller T, Ridder R, et al. Conjunctive p16INK4a testing significantly increases accuracy in diagnosing high-grade cervical intraepithelial neoplasia. Am J Clin Pathol. 2010;133(3):395-406. https://doi.org/10.1309/AJCPXSVCDZ3D5MZM
16.Gustinucci D, Passamonti B, Cesarini E, Butera D, Palmieri EA, Bulletti S, et al. Role of p16(INK4a) cytology testing as an adjunct to enhance the diagnostic specificity and accuracy in human papillomavirus-positive women within an organized cervical cancer screening program. Acta Cytol. 2012;56(5):506. https://doi.org/10.1159/000338979
17.von Knebel Doeberitz M, Reuschenbach M, Schmidt D, Bergeron C. Biomarkers for cervical cancer screening: the role of p16(INK4a) to highlight transforming HPV infections. Expert Rev Proteomics. 2012;9(2):149-63. https://doi.org/10.1586/epr.12.13
18.Ikenberg H, Bergeron C, Schmidt D, Griesser H, Alameda F, Angeloni C, et al. Screening for cervical cancer precursors with p16/Ki-67 dual-stained cytology: results of the PALMS study. J Natl Cancer Inst. 2013;105(20):1550. https://doi.org/10.1093/jnci/djt235
19.Petry KU, Schmidt D, Scherbring S, Luyten A, Reinecke-Lüthge A, Bergeron C, et al. Triaging Pap cytology negative, HPV positive cervical cancer screening results with p16/Ki-67 Dual-stained cytology. Gynecol Oncol. 2011;121(3):505. https://doi.org/10.1016/j.ygyno.2011.02.033
20.Wentzensen N, Bergeron C, Cas F, Vinokurova S, von Knebel Doeberitz M. Triage of women with ASCUS and LSIL cytology: use of qualitative assessment of p16INK4a positive cells to identify patients with high-grade cervical intraepithelial neoplasia. Cancer. 2007;111(1):58-66.
21.Toll AD, Kelly D, Maleki Z. Utility of P16 expression and Ki-67 proliferation index in ASCUS and ASC-H pap tests. Diagn Cytopathol. 2014;42(7):576. https://doi.org/10.1002/dc.23076
22.Atkins K. p16/Ki-67 dual-stain cytology in the triage of ASCUS and LSIL papanicolaou cytology: p16/Ki-67 in cervical papanicolaou Tests. Cancer cytopathology. 2011;119(3):145. https://doi.org/10.1002/cncy.20139
23.Schmidt D, Bergeron C, Denton KJ, Ridder R, Group ECCS. p16/ki-67 dual-stain cytology in the triage of ASCUS and LSIL papanicolaou cytology: results from the European equivocal or mildly abnormal Papanicolaou cytology study. Cancer cytopathology. 2011;119(3):158. https://doi.org/10.1002/cncy.20140
24.Nieh S, Chen SF, Chu TY, Lai HC, Lin YS, Fu E, et al. Is p16(INK4A) expression more useful than human papillomavirus test to determine the outcome of atypical squamous cells of undetermined significance-categorized Pap smear? A comparative analysis using abnormal cervical smears with follow-up biopsies. Gynecol Oncol. 2005;97(1):35-40.
25.Duncan L, Jacob S, Hubbard E. Evaluation of p16INK4a as a diagnostic tool in the triage of Pap smears demonstrating atypical squamous cells of undetermined significance. Cancer. 2008;114(1):34. https://doi.org/10.1002/cncr.23255
26.Wentzensen N, Schwartz L, Zuna RE, Smith K, Mathews C, Gold MA, et al. Performance of p16/Ki-67 immunostaining to detect cervical cancer precursors in a colposcopy referral population. Clin Cancer Res. 2012;18(15):4154. https://doi.org/10.1158/1078-0432.CCR-12-0270
27.Tsoumpou I, Arbyn M, Kyrgiou M, Wentzensen N, Koliopoulos G, Martin-Hirsch P, et al. p16(INK4a) immunostaining in cytological and histological specimens from the uterine cervix: a systematic review and meta-analysis. Cancer treatment reviews. 2009;35(3):210. https://doi.org/10.1016/j.ctrv.2008.10.005
28.Walts AE, Bose S. P16/Ki-67 Immunostaining is Useful in Stratification of Atypical Metaplastic Epithelium of the Cervix. Clin Med Pathol. 2008;1:35-42.
29.Regauer S, Reich O. CK17 and p16 expression patterns distinguish (atypical) immature squamous metaplasia from high-grade cervical intraepithelial neoplasia (CIN III). Histopathology. 2007;50(5):629-35.
30.Iaconis L, Hyjek E, Ellenson LH, Pirog EC. p16 and Ki-67 immunostaining in atypical immature squamous metaplasia of the uterine cervix: correlation with human papillomavirus detection. Arch Pathol Lab Med. 2007;131(9):1343-9.
31.Mittal K, Soslow R, McCluggage WG. Application of immunohistochemistry to gynecologic pathology. Arch Pathol Lab Med. 2008;132(3):402. https://doi.org/10.1043/1543-2165(2008)132[402:AOITGP]2.0.CO;2
32.Klaes R, Friedrich T, Spitkovsky D, Ridder R, Rudy W, Petry U, et al. Overexpression of p16(INK4A) as a specific marker for dysplastic and neoplastic epithelial cells of the cervix uteri. International journal of cancer. 2001;92(2):276-Epub 2001/04/06.
33.Trutnovsky G, Kolovetsiou-Kreiner V, Reich O. p16/Ki-67 dual-stained cytology testing may predict postpartum outcome in patients with abnormal papanicolaou cytology during pregnancy. Acta Cytol. 2014;58(3):293-6. https://doi.org/10.1159/000358817.
34.Solomon D, Davey D, Kurman R, Moriarty A, O'Connor D, Prey M, et al. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA. 2002;287(16):2114-9.
35.Denton KJ, Bergeron C, Klement P, Trunk MJ, Keller T, Ridder R, et al. The sensitivity and specificity of p16(INK4a) cytology vs HPV testing for detecting high-grade cervical disease in the triage of ASC-US and LSIL pap cytology results. Am J Clin Pathol. 2010;134(1):12. https://doi.org/10.1309/AJCP3CD9YKYFJDQL
36.del Pino M, Garcia S, Fusté V, Alonso I, Fusté P, Torné A, et al. Value of p16(INK4a) as a marker of progression/regression in cervical intraepithelial neoplasia grade Am J Obstet Gynecol. 2009;201(5):488.e1. https://doi.org/10.1016/j.ajog.2009.05.046
37.Yoshida T, Sano T, Kanuma T, Inoue H, Itoh T, Yazaki C, et al. Usefulness of CINtec® PLUS p16/Ki-67 double-staining in cytological screening of cervical cancer. Acta Cytol. 2011;55(5):413. https://doi.org/10.1159/000331047
38.Carozzi F, Confortini M, Dalla Palma P, Del Mistro A, Gillio-Tos A, De Marco L, et al. Use of p16-INK4A overexpression to increase the specificity of human papillomavirus testing: a nested substudy of the NTCC randomised controlled trial. The Lancet Oncology. 2008;9(10):937. https://doi.org/10.1016/S1470-2045(08)70208-0
39.Roelens J, Reuschenbach M, von Knebel Doeberitz M, Wentzensen N, Bergeron C, Arbyn M. p16INK4a immunocytochemistry versus human papillomavirus testing for triage of women with minor cytologic abnormalities: a systematic review and meta-analysis. Cancer cytopathology. 2012;120(5):294-307. https://doi.org/10.1002/cncy.21205
40.Reuschenbach M, Seiz M, von Knebel Doeberitz C, Vinokurova S, Duwe A, Ridder R, et al. Evaluation of cervical cone biopsies for coexpression of p16INK4a and Ki-67 in epithelial cells. International journal of cancer. 2012;130(2):388. https://doi.org/10.1002/ijc.26017
41.Klaes R, Benner A, Friedrich T, Ridder R, Herrington S, Jenkins D, et al. p16INK4a immunohistochemistry improves interobserver agreement in the diagnosis of cervical intraepithelial neoplasia. Am J Surg Pathol. 2002;26(11):1389-99.
42.Galgano MT, Castle PE, Atkins KA, Brix WK, Nassau SR, Stoler MH. Using biomarkers as objective standards in the diagnosis of cervical biopsies. Am J Surg Pathol. 2010;34(8):1077. https://doi.org/10.1097/PAS.0b013e3181e8b2c4
43.Sari Aslani F, Safaei A, Pourjabali M, Momtahan M. Evaluation of Ki67, p16 and CK17 Markers in Differentiating Cervical Intraepithelial Neoplasia and Benign Lesions. Iran J Med Sci. 2013;38(1):15-21.