Introduction
Clinically, pathologically and genetically, Frontotemporal Lobar Degeneration (FTLD) is a group of heterogeneous disorders that mainly affect the frontal and temporal lobes, usually in people younger than 65 years at the time of diagnosis (1). It represents the second most common form of dementia in people younger than 65 years, and diagnosis tends to be more difficult in older people (2). It has a prevalence of 10 to 30 cases per 100,000 people between 45 and 65 years of age (3). The most common clinical syndrome, accounting for more than half of cases, is the behavioral variant (4). However, the diagnosis of variants involving speech and language is becoming increasingly important. One of the less frequent language variants in terms of prevalence is agrammatic or non-fluent PPA, where a clinical presentation characterized by agrammatism, labored speech, paraphasias and difficulty in motor speech planning has been described (5).
Case A
The case corresponds to a 57 years-old patient, born and living in Bogota. He studied architecture and did a master’s degree in Economics, had his own company, lectured to entrepreneurs until 2016, and currently works as an independent architect. The patient says that for 16 years he has had anxiety symptoms, which have exacerbated, with excessive worries about trivial events, difficulty in planning and decision making.
In the last three years, affective symptoms have become more frequent, he worries about financial difficulties, has become more expressive and communicates his feelings and thoughts to his relatives more often, but participates less in social gatherings. His wife describes that the patient tells some facts out of context, with circumlocutions, and that he has difficulty in drawing conclusions and finding words; his verbal fluency has diminished, in association with ritualistic, stereotyped and perseverative behavior. In addition, he has difficulty in remembering some proper names, he must write down appointments and commitments more frequently, because it is difficult for him to organize his time. Currently, he persistently expresses subjective complaints regarding his memory (“he forgets where he leaves the keys,” “he forgets commitments and appointments”). A family member reports that they create strategies for planning activities (several weeks in advance they make a plan of the day-to-day activities), and with this they have noted an improvement in relation to the subjective complaints of forgetting appointments, commitments or objects. In problem solving, he remains typecast in one or two solutions, without allowing other possible solutions. In relation to language, they have noticed less verbal fluency, it is difficult for him to find words, especially in periods of anxiety, but also in quiet moments, when he narrates events centered on himself, excluding the other participants, slow processing, without paraphasia, omitting connectors in the grammatical composition (e.g. “home we go soon”) that sometimes it is possible to understand. No episodes of disorientation. Empathy and affection for other people has decreased. Associated with these changes in language, he has become much more interested in drawing. However, he has changed the pattern of his drawings and has been typecast into continuing to create paintings from a single photograph.
Discussion
Frontotemporal lobar degeneration is a clinically, genetically, and pathologically heterogeneous neurodegenerative disorder that causes selective neuronal loss and gliosis of the frontal and temporal lobes of the brain. Frontotemporal lobar degeneration is characterized by neuronal loss, gliosis, and microvacuolar changes of frontal lobes, anterior temporal lobes, anterior cingulate cortex, and insular cortex. Subtypes are associated with characteristic patterns of abnormal protein deposition (6). Initial changes occur in the anterior cingulate cortex, fronto-insular cortex, orbitofrontal cortex, and cingulate-frontal transitional zones. These regions have von Economo neurons and fork cells in layer 5 of the cortex, which are thought to play a central part in the integration of cortical and subcortical networks, and degenerate very early in behavioural-variant frontotemporal dementia (7). Either the microtubule-associated protein tau, the TAR DNA-binding protein with molecular weight 43 kDa (TDP-43), or the fused-in-sarcoma (FUS) protein account for nearly all cases of frontotemporal lobar degeneration. The corresponding pathological subtypes of frontotemporal lobar degeneration are frontotemporal lobar degeneration-tau, frontotemporal lobar degeneration-TDP, and frontotemporal lobar degeneration-FUS (6).
As mentioned previously, the diagnosis of PPA is based on objective deterioration of language and speech, while other cognitive domains such as episodic and topographic memory, and constructive praxis are relatively well preserved (8); therefore, in the initial stages the affectation of daily life activities is totally attributable to language problems (5). It should be noted that, although many patients do not have memory impairment, those who do may be misdiagnosed with Alzheimer’s disease with left hemisphere involvement, where language symptoms are more prominent (3).
Globally, a highly variable incidence of non-fluent PPA has been described, ranging from 2.2 to 3.5 cases per 100,000 people. 45% of FTLD cases are due to PPA, and less than half of the cases are due to non-fluent PPAg (9). Some studies have described a much higher prevalence of non-fluent PPA in females, while others report no difference between the two genders (4, 9); some have even suggested that the age of onset may be much later than in other forms of FTLD (4). One study showed an average age of onset of 63 years for non-fluent PPA vs. 57.5 years for FTLD patients and 59.3 years for the semantic PPA variant (4), with an average survival of approximately 7 years for non-fluent PPA (5).
Clinically, patients with non-fluent PPA present with low quality labored speech. Some studies have shown that the average speech rate produced by nfvPPA patients is about 45 words per minute compared to 140 words per minute in healthy controls. This group of patients shows slow speech, with effort and grammatical errors and/or apraxia of speech, that is, in the planning and motor sequencing of the lips, tongue and breathing necessary to articulate speech (4). They usually have difficulty finding words, speak in short phrases, omit articles (a, the) and show pronunciation difficulties, similar to what is observed in Broca’s aphasia (9). Patients may have aphemia, but in the early stages they can communicate correctly in writing (9). Anatomically, the correlation with the clinical picture is explained by the involvement of Broadman’s area 44 and 45 (Broca’s area) in the lower left frontal convolution and the anterior insula (5).
To diagnose these disorders, it is essential to evaluate language along with other domains. Different tests can be used such as the verbal fluency test, the Boston Naming Test, and the Boston Diagnostic Aphasia Battery, among others. Generally, in early stages it is common for the patient to have difficulty finding words, but frank anomia is rare. Comprehension usually remains intact, except for longer complex sentences. The executive function may show subtle dysfunction, but memory and visuospatial skills remain relatively free early in the course of the disease (3).
For a patient to meet the clinical criteria for non-fluent variant primary progressive aphasia, they must have a speech with apraxia or motor speech agrammatism as well as supporting features (Table 1).
Table 1.
Features that distinguish dementia of the frontal type from dementia of the Alzheimer’s type in the early stage of the disease
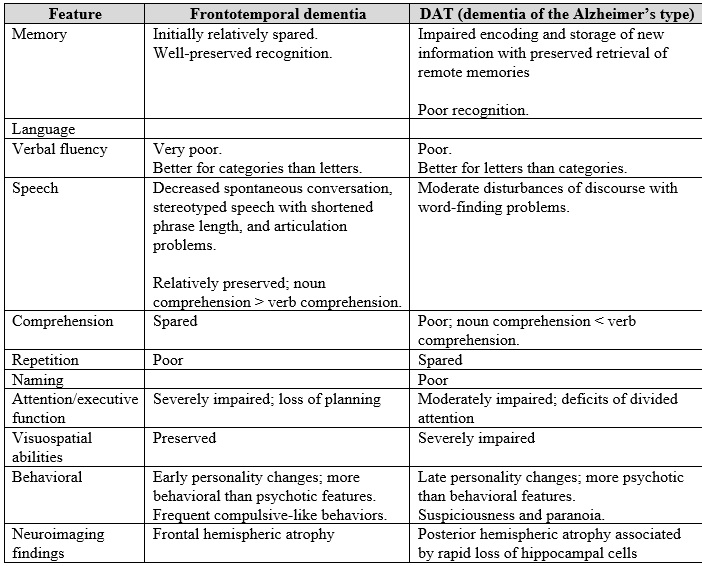
Source: adapted from Harciarek M, Jodzio K. Neuropsychological differences between frontotemporal dementia and Alzheimer’s disease: a review. Neuropsychol Rev. 2005 Sep;15(3):131–45
Neuroimaging studies for non-fluent variant primary progressive aphasia may be normal at the time of presentation. However, the changes found are consistent with atrophy in the brain MRI or hypometabolism in the PET or SPECT scan in the lower left frontal convolution near the perisilvian area and involves the Broca’s area (10).
Treatment is symptomatic. The FDA has not approved drugs for frontotemporal degeneration. The use of acetylcholinesterase inhibitors has been shown to worsen symptoms in this group of patients (10, 11). In a double-blind, placebo-controlled trial, the use of memantine in patients with frontotemporal degeneration showed no benefit on behavior or cognition (11, 12).
Behavioral symptoms in frontotemporal degeneration variants can be managed with selective serotonin inhibitors. Atypical antipsychotic drugs can be used in frontotemporal degeneration, but caution should be exercised, given the vulnerability to extrapyramidal side effects (12). The use of non-pharmacological therapies that include behavioral, environmental and physical techniques, in addition to caregiver education, may help minimize or redirect unwanted behaviors, while language deficits may be slightly improved with speech therapy in patients with variant primary progressive aphasia (10, 13).
Conclusion
Although the agrammatic or non-fluent variant of PPA is a rare entity, with limitations in its early diagnosis and treatment, it should be considered within the differential diagnosis of young people with affective and language symptoms, since its early diagnosis can improve the quality of life of the patient and their family.
Conflicts of interests
We declare that we are aware of the journal’s conflict of interest policy. The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our case report has the ethical approval of the Institutional Committee to report individual cases or series of cases.
Financial disclosure
We declare that this research work is original and is not being evaluated by any other scientific journal. We declare that the corresponding ethical authorities have been taken into account to carry out this research work, all authors agree with the present version of the manuscript and there are no conflicts of interest. We declare that no financial support or sponsors were required to carry out the research and / or the preparation of the article.
References
1. Mann DMA, Snowden JS. Frontotemporal lobar degeneration: Pathogenesis, pathology and pathways to phenotype. Brain Pathol. 2017 Nov;27(6):723–36. https://doi.org/10.1111/bpa.12486
2. Bürger K, Arzberger T, Stephan J, Levin J, Edbauer D. Pathomechanismen und klinische Aspekte der frontotemporalen Lobärdegeneration. Nervenarzt. 2017 Feb 20;88(2):163–72. https://doi.org/10.1007/s00115-016-0259-x
3. Bennion Callister J, Pickering-Brown SM. Pathogenesis/genetics of frontotemporal dementia and how it relates to ALS. Exp Neurol. 2014 Dec;262 Pt B:84–90. https://doi.org/10.1016/j.expneurol.2014.06.001
4. Ioannidis P, Konstantinopoulou E, Maiovis P, Karacostas D. The frontotemporal dementias in a tertiary referral center: Classification and demographic characteristics in a series of 232 cases. J Neurol Sci. 2012 Jul 15;318(1–2):171–3. https://doi.org/10.1016/j.jns.2012.04.002
5. Matías-Guiu JA, García-Ramos R. Afasia progresiva primaria: del síndrome a la enfermedad. Neurología. 2013 Jul;28(6):366–74. https://doi.org/10.1016/j.nrl.2012.04.003
6. Bang J, Spina S, Miller BL. Frontotemporal dementia. Lancet (London, England). 2015 Oct 24;386(10004):1672–82. https://doi.org/10.1016/S0140-6736(15)00461-4
7. Kim E-J, Sidhu M, Gaus SE, Huang EJ, Hof PR, Miller BL, et al. Selective frontoinsular von Economo neuron and fork cell loss in early behavioral variant frontotemporal dementia. Cereb Cortex. 2012 Feb;22(2):251–9.
8. Ioannidis P, Konstantinopoulou E, Maiovis P, Karacostas D. The frontotemporal dementias in a tertiary referral center: classification and demographic characteristics in a series of 232 cases. J Neurol Sci. 2012 Jul 15;318(1–2):171–3. https://doi.org/10.1016/j.jns.2012.04.002
9. Grossman M. The non-fluent/agrammatic variant of primary progressive aphasia. Lancet Neurol. 2012 Jun;11(6):545–55. https://doi.org/10.1016/S1474-4422(12)70099-6
10. Boxer AL, Knopman DS, Kaufer DI, Grossman M, Onyike C, Graf-Radford N, et al. Memantine in patients with frontotemporal lobar degeneration: a multicentre, randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2013 Feb ;12(2):149–56. https://doi.org/10.1016/S1474-4422(12)70320-4
11. Pijnenburg YAL, Sampson EL, Harvey RJ, Fox NC, Rossor MN. Vulnerability to neuroleptic side effects in frontotemporal lobar degeneration. Int J Geriatr Psychiatry. 2003 Jan;18(1):67–72. https://doi.org/10.1002/gps.774
12. Merrilees J. A Model for management of behavioral symptoms in frontotemporal lobar degeneration. Alzheimer Dis Assoc Disord. 2007 Oct;21(4):S64–9. https://doi.org/10.1097/WAD.0b013e31815bf774
13. Kortte KB, Rogalski EJ. Behavioural interventions for enhancing life participation in behavioural variant frontotemporal dementia and primary progressive aphasia. Int Rev Psychiatry. 2013 Apr 24 [cited 2019 Aug 6];25(2):237–45. https://doi.org/10.3109/09540261.2012.751017
Author notes
a Corresponding author: arizacarlos@javeriana.edu.co
Additional information
How to cite: Ariza-Galindo CJ, Santacruz-Escudero JM, Lozano-Rengifo MJ, Segura-Valencia AI. Frontotemporal lobar degeneration – agrammatic primary progressive aphasia: case report. Univ. Med. 2021;62(1). https://doi.org/10.11144/Javeriana.umed62-1.apha