Introduction
The prescription of a drug approved by international regulatory agencies, such as the European Medicines Agency (EMA) (1) or the Food and Drug Administration (FDA) (2), and in Colombia, by the Instituto Nacional de Vigilancia de Medicamentos y Alimentos (Invima) (3), certifies that a rigorous evaluation has been carried out and that the benefits have proven to outweigh the risks of the treatment for a particular indication. Eguale et al. (4) demonstrated that the use of unapproved drugs, or drugs with weak evidence, is associated with a higher frequency of adverse reactions (4), which leads to an increase in the number of days of stay and costs, which has a collateral impact on the finances of the healthcare system (5).
Human intravenous immunoglobulin (IVIg) is a high-cost healthcare technology that has demonstrated a positive impact on the management of different acute and chronic diseases (6-8). Different regulatory agencies agree on approving its use in pathologies such as Guillain-Barré syndrome, immune thrombocytopenic purpura, repeated infections in HIV patients, and common variable immunodeficiency, among others (9,10). The number of approved indications changes according to the regulatory agency: nine for the FDA (11), 10 for the EMA (9), and 12 for Invima (12,13). Despite this, its use in clinical practice has been extended to 24 unapproved indications (6,14,15).
In Colombia, studies describing the use of IVIgI have not yet been published. This information would be essential to exploring both the pattern of use and the determinants that influence its use, especially in the context of its application under indications approved by regulatory agencies (14-16). The objective of the present study was to describe the pattern of use, the determinants of use, and the costs of IVIg administration in a high-complexity institution in Bogotá (Colombia), especially in indications not approved by the FDA, EMA, and Invima regulatory agencies.
Methodology
We conducted a drug utilization study according to the methodology proposed by the World Health Organization (17,18). This can be described as a cross-sectional observational trial in which variables such as the pattern, quality, and determinants of IVIg use are described.
We included all patients who were prescribed and administered IVIg in the period from January 1, 2013 to January 1, 2018, in a general hospital that treats highly complex pathologies in the city of Bogota (Colombia), without age limits. Patients who did not complete the prescribed IVIg scheme were excluded, either because the dose was not completed, due to patient death, or because of a referral to another institution.
The patients who received IVIg were identified with the DI-SEARCH tool (19), based on the information contained in the electronic medical records and structured data, such as the diagnoses of the International Classification of Diseases (ICD-10) as well as based on narrative data recorded in free text.
For each patient, age, weight, gender, comorbidities, the number of IVIg prescriptions, the indication for which it was prescribed, the completion of the informed consent form for patients who had IVIg administered in an unapproved (off-label) indication, its cost, and the adverse reactions described by the prescribing physician were evaluated. Data were collected from the patients' electronic records. The medical records were evaluated independently by two investigators (DD and NC). Differences between the evaluators were solved by consensus.
The cost of the IVIg was taken from the database of the Integrated Social Protection Information System, which corresponds to the information system of the Colombian Ministry of Health and Social Protection, where data on supply and demand of health services, quality of services, insurance, financing, and social promotion are stored (20). Specifically, the costs are recorded in the "Drug Price Thermometer" application. The cost of IVIg corresponds to the institutional channel price reported by pharmaceutical laboratories (21). Given that the cost of the drug encompasses both its price and the expenses associated with its administration (such as the use of infusion rooms, hospitalization, and medical personnel for administration), and considering that administration costs are uniform across all applications, the distinguishing factor lies in the price of the drug itself. For this reason, in our report, we have only considered the price of the drug as the central element.
Quantitative variables were evaluated using the Kolmogorov-Smirnov test, and depending on the distribution, the mean and standard deviation, or median and interquartile range, were reported. Categorical variables are presented as absolute frequencies and percentages. For the comparison of costs between indications approved and those not approved by the regulatory agencies, the Mann-Whitney U test was used. A p value < 0.05 was considered statistically significant. Data processing and analysis were performed in SPSS version 18.
The institution's Ethics Committee approved the research protocol. No sensitive patient information was collected.
Results
A total of 66 patients were included in the analysis. The demographic characteristics of the patients are presented in Table 1. Twelve (7.6%) of them were older than 65 years, and 56 (35.8%) were patients younger than 15 years. Out of the group analyzed, 56 patients received IVIg for FDA- and EMA-approved indications. The most frequent approved indications for use were Guillain-Barré syndrome (50%) and immune thrombocytopenic purpura (22.7%). Ten patients received the drug for non-approved indications, such as polymyositis or dermatopolymyositis (10.6%). The most frequent comorbidities were human immunodeficiency virus (HIV), type 2 diabetes mellitus, and hypothyroidism. Only one pregnant woman received IVIg, who was in the 35th week of gestation and the indication in this case was immune thrombocytopenic purpura.
Table 1
Demographic characteristics of patients (n = 66)

SD: standard deviation.
Approximately half of the prescriptions were made by physicians in training. No records of informed consent prior to IVIg administration were found for unapproved indications.
When evaluating IVIg administration by the number of doses administered, 156 individual events were found. The female gender received 85 doses. Table 2 discriminates the indications for which each dose of IVIg was prescribed, according to its approval by the regulatory agencies. Patients with polymyositis and dermatomyositis received 23.7% of all indications, with an average of 5.3 administrations per patient, outperforming other indications approved by regulatory agencies. In total, 28.2% of prescriptions were for indications not approved by the FDA or EMA. This proportion was slightly different for the indications approved by Invima (28.8%), because there are differences in the approval for two indications: multifocal motor neuropathy and Wiskott-Aldrich syndrome (Table 2).
Table 2
Indications for administration of intravenous human immunoglobulin, as approved by regulatory agencies
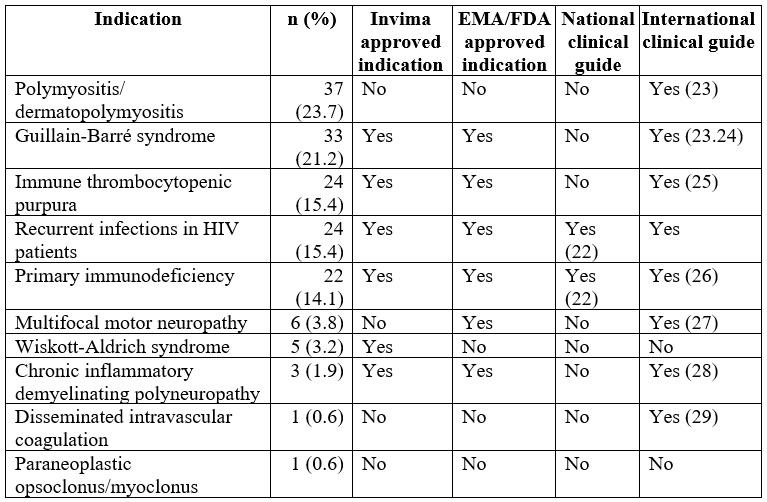
EMA: European Medicine Agency; Invima: Instituto Nacional de Vigilancia de Medicamentos y Alimentos; FDA: Food and Drug Administration.
There are international guidelines supporting the use of IVIg in 8 of the 10 indications studied, and out of these, the two pathologies found are Wiskott-Aldrich syndrome and paraneoplastic opsoclonus/myoclonus. Only two Colombian national guidelines have been developed recommending its use.
Table 3 shows the drug costs. The cost of administration (administration rooms, medical and nursing staff, supplies, among others) is not included. The average cost (in Colombian pesos [COP]) of using the drug in the non-approved indications exceeded that of the indications approved by Invima (16,713,213 COP vs. 8,383,855 COP; p <0.001). The result was similar when comparing cost by FDA and EMA approval (13,881,994 COP vs. 9,570,488 COP; p < 0.015).
Table 3
Intravenous human immunoglobulin administration costs
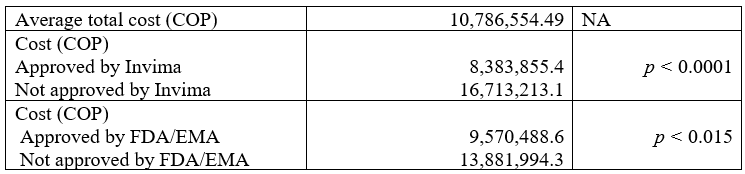
COP: Colombian pesos; EMA: European Medicine Agency; Invima: Instituto Nacional de Vigilancia de Medicamentos y Alimentos; FDA: Food and Drug Administration.
Adverse reactions were identified in four patients, with one classified as serious (bradycardia) and three non-serious (headache). Causality was assessed for the serious adverse reaction using the Naranjo scale, with a probable result (of 5 points). This adverse reaction occurred in one patient who received IVIg for an indication approved by the three regulatory agencies. Due to the appearance of bradycardia, IVIg administration was discontinued in this patient. In the non-serious adverse reactions, there was no discontinuation of the drug.
Discussion
The present study is the first to evaluate the pattern of use, determinants of use, and costs of IVIg in Colombia, using the methodology proposed by the World Health Organization for a drug utilization study (17). We document that there is a high proportion of prescriptions for indications not approved by the regulatory agencies, and that such administrations have a significantly higher cost.
As it is an immunomodulatory and humoral immunity replacement therapy, IVIg can be prescribed for multiple indications, including several not approved by the regulatory agencies due to the lack of strong scientific evidence (14-16,30,31). This problematic documented in our study has also been evaluated and found in several places in the world (14-16). In Spain, Ruiz-Antorán et al. (14) conducted a study in 13 Spanish hospitals in which the use of IVIg was evaluated, and showed that 16% of patients received IVIg for non-approved indications.
We have also recorded that non-approved indications have a significantly higher cost compared to approved indications. This finding is in agreement with similar research carried out by Ruiz-Antorán et al. (14), who documented that the average cost of approved indications was 2626.2 euros while that of non-approved indications amounted to 3555.8 euros on average. Although the increase in cost may be the result of multiple variables, in our study we identified in particular that this is higher in non-approved indications, possibly due to the need for higher dosing in this setting.
It is worth mentioning that adverse reactions did not play a determining role in the price increase. This was evidenced by the fact that only one serious adverse reaction (bradycardia) was recorded, which led to the suspension of IVIg administration without requiring additional interventions.
In our study, polymyositis and dermatomyositis were the main indication for IVIg administrations over approved indications such as immune thrombocytopenic purpura, Guillain-Barré syndrome, and recurrent infections in HIV patients (6,22,23,32,33). The creation of patient registries for low-prevalence diseases such as polymyositis and dermatomyositis is shown to be a tool that will potentially help us generate evidence on the use of IVIg in patients who have been refractory to management with steroids, azathioprine, and rituximab (34).
In addition, we found differences in the number of approved indications among the different regulatory agencies (FDA, EMA, and Invima), a fact that calls for harmonization in drug regulation.
In our study, the rate of serious and non-serious adverse events was low; however, previous studies have documented that the use of off-label drugs increases costs, in addition to serious and non-serious adverse drug reactions (4). This problematic has also been evaluated in oncology, where antineoplastic drugs are prescribed as compassionate drugs, despite the lack of adequate evidence and, very often, without the informed consent of the patient, who receives a drug that has not been adequately studied and has a high potential for toxicity (35).
The main limitation of our study is that it reports the use of IVIg in only one health institution. Although its use in a high-complexity hospital does not necessarily reflect the reality of a country, it can be representative of the reality of national and regional high-complexity institutions. A second limitation is that the number of prescriptions is not very high, and despite this, the difference in costs for non-approved indications was clearly significant.
Although the use of drugs not approved by regulatory agencies does not mean that medical decisions are wrong, it does identify gaps in knowledge, which we should seek to fill by promoting research into therapeutic alternatives, fostering the creation of scientific consensus on the treatment of low-prevalence diseases, and favoring the harmonization of local, national, and international regulations.
In summary, our study has revealed that, despite the fact that IVIg prescriptions are markedly lower compared to non-approved indications, the cost associated with the latter is considerably higher. This imbalance is largely attributed to the need to administer higher doses of the drug in unapproved indications. These results underscore the importance of undertaking future research or evidence gathering to support or rule out the use of these off-label indications, as they carry significant costs to healthcare systems.
In addition, our study highlights how regulatory agencies can grant approvals in a differentiated manner, which in turn calls for a deeper evaluation of the methodologies employed in drug approval. It is essential to seek global harmonization of these criteria to establish coherent and consistent standards at the international level.
Funding
This work did not receive any external financial support.
Conflict of interests
The authors have no conflicts of interest to declare.
Acknowledgments
Special thanks to Albert Figueras-Suñé, PhD, thesis director, for the application for the title of Master in Pharmacovigilance and Pharmacoepidemiology of the European Program in Pharmacovigilance and Pharmacoepidemiology (EU2P).
References
1. European Medicines Agency [Internet]. Available from: https://www.ema.europa.eu/en
2. U.S. Food and Drug Administration [Internet]. Available from: https://www.fda.gov/
3. Instituto Nacional de Vigilancia de Medicamentos y Alimentos [Internet]. Available from: https://www.invima.gov.co/
4. Eguale T, Buckeridge DL, Verma A, Winslade NE, Benedetti A, Hanley JA, et al. Association of off-label drug use and adverse drug events in an adult population. JAMA Intern Med. 2016 Jan 1;176(1):55-63. https://doi.org/10.1001/jamainternmed.2015.6058
5. Solarte KG, Benavides Acosta FP, Rosales Jiménez R. Costos de la enfermedad crónica no transmisible: la realidad colombiana. Rev Cienc Salud. 2016;14(1):103-14. https://doi.org/10.12804/revsalud14.01.2016.09
6. Perez EE, Orange JS, Bonilla F, Chinen J, Chinn IK, Dorsey M, et al. Update on the use of immunoglobulin in human disease: a review of evidence. J Allergy Clin Immunol. 2017;139(3):S1-46. https://doi.org/10.1016/j.jaci.2016.09.023
7. Shabaninejad H, Asgharzadeh A, Rezaei N, Rezapour A. Estimating the cost of immunoglobulin replacement therapy in primary immunodeficiency patients. Iran J Pediatr. 2017;27(4):e6294. https://doi.org/10.5812/ijp.6294
8. Barnett C, Wilson G, Barth D, Katzberg HD, Bril V. Changes in quality of life scores with intravenous immunoglobulin or plasmapheresis in patients with myasthenia gravis [Internet]. J Neurol Neurosurg Psychiatry. 2013 Jan 1;84(1):94-7. Available from: https://jnnp.bmj.com/content/84/1/94.citation-tools
9. European Medicine Agency. Privigen [Internet]. Available from: https://www.ema.europa.eu/medicines/human/EPAR/privigen
10. Invima. Resolución 2013032232/2013, del 30 de octubre, registro sanitario inmunoglobina humana normal para administración IV.
11. U.S. Food and Drug Administration. Approved blood products - immune globulins [Internet]. Available from: https://www.fda.gov/vaccines-blood-biologics/approved-blood-products/immune-globulins
12. Invima. Inmunoglobulina humana normal para administración intravascular [Internet]. Available from: http://medicamentosaunclic.gov.co/Consultas/frmBusquedasIfrm.aspx?idPpio=1003
13. Invima CR. Sesión ordinaria Sala Especializada de Medicamentos y Productos Biologicos. Acta 8. 2017; p. 1-544.
14. Ruiz-Antorán B, Escasany AA, Ferraz AV, Carreras ID, Riba N, Escudero SM, et al. Use of non-specific intravenous human immunoglobulins in Spanish hospitals; need for a hospital protocol. Eur J Clin Pharmacol. 2010;66(6):633-41.
15. Alangari AA, Abutaleb MH, Albarraq AA, Al-Dhowailie AA. Intravenous immunoglobulin utilization in a tertiary care teaching hospital in Saudi Arabia. Saudi Med J. 2008 Jul;29(7):975-9.
16. Darabi K, Abdel-Wahab O, Dzik WH. Current usage of intravenous immune globulin and the rationale behind it: The Massachusetts General Hospital data and a review of the literature. Transfusion. 2006;46(5):741-53.
17. World Health Organization. Introduction to drug utilization research [Internet]. 2003. Available from: https://iris.who.int/handle/10665/42627?&locale-attribute=de
18. Shalini S, Ravichandran V, Bk M, Sk D, Saraswathi R. Shalini S. et al. Drug utilization studies-an overview drug utilization studies-an overview. Int J Phamaceut Sci Nanotechnol. 2010;3(1). https://doi.org/10.37285/ijpsn.2010.3.1.2
19. Quimbaya AP, González RA, Bohórquez WR, Muñoz O, García OM, Londoño D. A systemic, participative design of decision support services for clinical research. Int J Inf Technol Syst Approach. 2014 Jun;7(2):20-40.
20. Ministerio de Salud y Protección Social. Termómetro de precios de medicamentos [Internet]. Available from: https://www.minsalud.gov.co/salud/MT/Paginas/termometro-de-precios.aspx
21. Sistema Integrado de Información de la Protección Social (SISPRO) [Internet]. Available from: https://www.sispro.gov.co/Pages/Home.aspx
22. Wang DX, Shu XM, Tian XL, Chen F, Zu N, Ma L, et al. Intravenous immunoglobulin therapy in adult patients with polymyositis/dermatomyositis: a systematic literature review. Clin Rheumatol. 2012 May 26;31(5):801-6. 25
23. Patwa HS, Chaudhry V, Katzberg H, Rae-Grant AD, So YT. Evidence-based guideline: intravenous immunoglobulin in the treatment of neuromuscular disorders: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology. 2012 Mar 27;78(13):1009-15. https://doi.org/10.1212/WNL.0b013e31824de293
24. Meena AK, Khadilkar S V, Murthy JMK. Treatment guidelines for Guillain-Barré Syndrome. Ann Indian Acad Neurol. 2011 Jul;14(Suppl 1):S73-81. https://doi.org/10.4103/0972-2327.83087
25. Neunert C, Lim W, Crowther M, Cohen A, Solberg L, Crowther MA, et al. The American Society of Hematology 2011 evidence-based practice guideline for immune thrombocytopenia. Blood. 2011 Apr 21;117(16):4190-207. https://doi.org/10.1182/blood-2010-08-302984
26. Yong PL, Boyle J, Ballow M, Boyle M, Berger M, Bleesing J, et al. Use of intravenous immunoglobulin and adjunctive therapies in the treatment of primary immunodeficiencies. Clin Immunol. 2010 May;135(2):255-63. https://doi.org/10.1016/j.clim.2009.10.003
27. Guidelines PNSMMN. European Federation of Neurological Societies / Peripheral Nerve Society Guideline on management of multifocal motor neuropathy. Report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society. 2006;8:1-8.
28. Van den Bergh PYK, Hadden RDM, Bouche P, Cornblath DR, Hahn A, Illa I, et al. European Federation of Neurological Societies/Peripheral Nerve Society Guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European Federation of Neurological Societies and the Peripher. Eur J Neurol. 2010;17(3):356-63. 35
29. Wada H, Thachil J, Di Nisio M, Mathew P, Kurosawa S, Gando S, et al. Guidance for diagnosis and treatment of disseminated intravascular coagulation from harmonization of the recommendations from three guidelines. J Thromb Haemost. 2013;11:761-8. https://doi.org/10.1111/jth.12155
30. Dorado García M. Uso de fármacos fuera de indicación en patología dual, necesidad clínica. Rev Patol Dual. 2014;1(3):1-5.
31. Czernik A, Toosi S, Bystryn J-C, Grando SA. Intravenous immunoglobulin in the treatment of autoimmune bullous dermatoses: an update. Autoimmunity. 2012 Feb 19;45(1):111-8. https://doi.org/10.3109/08916934.2011.606452
32. Rosier C, Graveline N, Lacour A, Antoine J-C, Camdessanché J-P. IGIV for chronic inflammatory demyelinating polyneuropathies and multifocal motor neuropathies treatment in France: are daily practices in accordance with guidelines? Eur J Neurol. 2018 Oct 16. https://doi.org/10.1111/ene.13841
33. Galimberti F, Kooistra L, Li Y, Chatterjee S, Fernandez AP. Intravenous immunoglobulin is an effective treatment for refractory cutaneous dermatomyositis. Clin Exp Dermatol. 2018 Jun 1.
34. McGettigan P, Alonso Olmo C, Plueschke K, Castillon M, Nogueras Zondag D, Bahri P, et al. Patient registries: an underused resource for medicines evaluation operational proposals for increasing the use of patient registries in regulatory assessments. Drug Saf. 2019;42:1343-51. https://doi.org/10.1007/s40264-019-00848-9
35. Saiyed MM, Ong PS, Chew L. Off-label drug use in oncology: a systematic review of literature. J Clin Pharm Ther. 2017;42(3):251-8. https://doi.org/10.1111/jcpt.12507
Author notes
a Correspondence author: nfcorrea@husi.org.co
Additional information
How to cite: Correa González NF, Díaz-Cortés D, Muñoz-Velandia OM. Intravenous immunoglobulin costs are higher for off-label indications in a high-complexity hospital in Colombia. Univ. Med. 2023;64(4). https://doi.org/10.11144/Javeriana.umed64-4.offl