INTRODUCTION
The oral cavity harbors a variety of microorganisms ranging from spores, bacteria, fungi, and viruses to protozoa. It contains about six billion bacteria, making it one of the most populated habitats in the human body (1,2,3). For this reason, dental practice is considered to carry a high risk of transmission of pathogens and cross-infection that can cause anything from a simple flu to diseases such as pneumonia, hepatitis B, and tuberculosis (4-5). In this sense, the proper disinfection and sterilization of dental instruments and handpieces is very important. These actions seek to avoid spreading infectious diseases from patient to patient and even among professionals (6,7,8,9). The Pan American Health Organization considers that instruments are sterile when they are free of living microorganisms (10).
Currently, moist heat using steam (autoclave) and dry heat (sterilizer) represent the most frequent procedures in dental offices (11). Currently, the Biosafety Manual for establishments, prepared by the Ministry of Public Health of Ecuador, suggests moist heat at 121 °C for 30 minutes. It also recommends to considering factors that affect sterilization such as incomplete removal of air in the sterilizer, superheated steam, and inadequate preparation of the material (6,12).
As mentioned above, the disinfection and sterilization of instruments has become more important because, during dental procedures, the staff is continuously in contact with body fluids such as saliva and blood. At the present time, due to the Covid-19 pandemic, the risk of contagion of infections is even higher (13). Such a virus has been shown to survive on steel and aluminum surfaces for 4 hours to 5 days. Even on plastic surfaces, the virus can live up to 9 days. However, the virus becomes infective when exposed to temperatures close to 75 ºC for 30 minutes (14).
One challenge is that if sterilization protocols are not followed or preventative maintenance on autoclaves is not performed, sterilization is ineffective. There is little literature on the effectiveness of sterilization processes in public or private clinics in cities of Ecuador, for example, the city of Machala. Consequently, the present study was based on the following research question: Are the autoclaves used in private clinics in the city of Machala, Ecuador effective?
The verification of the sterilization processes will allow oral health professionals to know the effectiveness of the autoclaves, carry out corrective actions, and reduce the possibility of cross-infection and infectious diseases. A strategy to verify said effectiveness are chemical indicators (15,16,17,18). One of them, the 3M® SteriGage® 1243, integrates all the critical parameters of steam sterilization: temperature, time, and steam. In addition, it can be used for all steam cycles at 121 ºC and 132 ºC (19). Thus, the main purpose of the study was to verify the effectiveness of autoclaves, through the use of chemical indicators, in private practice dental offices in the city of Machala, Ecuador. This study will serve as a starting point for future research on this topic.
MATERIALS AND METHODS
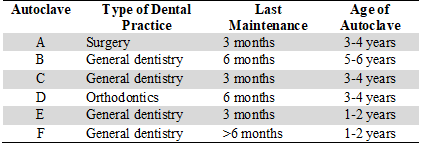
It was a descriptive study with an observational design (20) and a non-probabilistic sampling, which was approved by the Ethics Committee for Research on Human Beings (CEISH) of the Catholic University of Cuenca, Ecuador. It specifies that the research does not generate bioethical conflicts or risk as there is no contact with human beings. Invitations to participate in the study were sent to private practices, which were accepted by six practices. The owners of the private practices signed informed consents to participate and their identity was kept confidential. The autoclaves of these clinics were identified with the letters A, B, C, D, E, and F (Table 1). Only autoclaves that were currently active were included.
TABLE 1
Characteristics of Autoclaves
Source: the authors.
The variables analyzed were type of office, date of last maintenance, and age of the autoclave. In some of the dental specialties there is greater exposure to microorganisms. On the other hand, the time elapsed after the last maintenance and the age of the autoclave can influence its effectiveness, considering that preventive maintenance must be carried out twice a year.
To measure the effectiveness of the autoclaves, the 3M® SteriGage® 1243 integrating indicator was used. The measurement with the integrators was carried out twice a week for 5 weeks, which generated 10 measurements per autoclave, for a total of 60 samples. The indicators were applied for five weeks to avoid false positives.
A protocol for the use of the indicators was established. First, the dental instruments were manually cleaned, immersing them in an enzymatic detergent, and then each instrument was individually washed with a long-handled brush, as suggested by the Biosafety Manual of the Ministry of Health of Ecuador. Once washed, the instruments were packed in bags to be sterilized, ensuring that they were airtight and sealed. 50 % loads were applied as recommended by Lara Van der Linde, et al. (21), to avoid forcing situations that would bias the study due to excess load. The indicator was placed in the middle part of the tray; the position was a determining factor in reducing the risk of false positives. In this way, it allowed to monitor the effectiveness of the procedure in several points of the autoclave; thus, an effective process was guaranteed.
3M® SteriGage® Indicator 1243 consists of a solid, temperature-sensitive chemical that migrates through paper according to sterilization conditions. The result was analyzed through a window such as “Rejected” or “Accepted.” Accepted means the absence of bacterial growth.
The sterilization process was carried out in accordance with the Biosafety Manual for establishments prepared by the Ministry of Health of Ecuador, which recommends using the autoclave at a temperature of 121 °C for 30 minutes in order to achieve an effective sterilization cycle. When a cycle with an unaccepted indicator was reported, it was recommended to repeat the process, increasing the temperature to 132 °C for 15 minutes (12). Despite this, if the error persisted, the autoclave underwent maintenance, which improved its effectiveness. The quantification of the verification recommended by the Ministry of Health of Ecuador was as follows (12):
The data were processed using the statistical program RStudio version 2.15. Descriptive data for each variable were calculated. The values obtained were analyzed using Fisher's exact test to determine if two dichotomous variables were associated. Statistical significance was established when p = 1 (alpha error = 5 %).
RESULTS
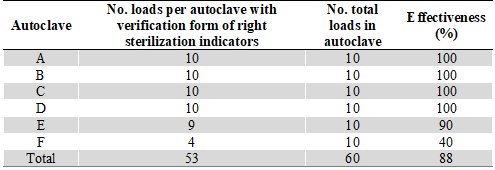
Of the groups analyzed per autoclave, A, B, C, and D showed 100 % effectiveness in sterilization. However, groups E and F had a lower effectiveness, with 90 % and 40 %, respectively (Table 2). According to Fisher's exact test, there was no significance between the effectiveness of sterilization of dental instruments and the variables type of office, maintenance time, and age of the autoclave (Table 3).
TABLE 2
Percentage of Dental Instruments’ Sterilization Effectiveness
Source: the authors.
TABLE 3
Descriptive Data and Fisher’s Test for each Type of Variable
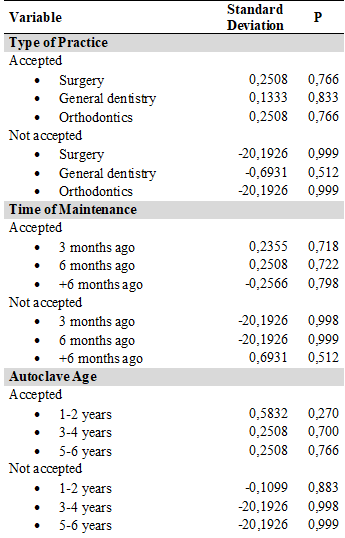
P = 1. Confidence interval 95 %. Minimum 0.182; maximum 2.47. ODDS Ratio: 1.13.
Association measures: Likelihood Ratio Chi square: 0.87. Likelihood Ratio Pearson: 0.25. Degrees of Freedom: 9. Phi: 0,029. Contingency coefficient COEF: 0,029; Kramer’s V: 0,029. Source: the authors.
DISCUSSION
No studies have been found in which autoclave sterilization has been evaluated in dental offices in the city of Machala, Ecuador. However, it is known that the sterilization protocol established by the Ministry of Health is used in most dental offices throughout the country. With this precedent, we sought to evaluate this protocol using 3M® SteriGage® 1243 integrative indicators in a non-probabilistic sample of autoclaves from private practices (12). It was found that 88.33 % of the six autoclaves/sterilization processes analyzed were effective, of which four autoclaves effective in 100 %, while there were less than 50 % of favorable readings in one autoclave; likewise, readings from an autoclave showed a 10 % error.
In the last two cases, they were autoclaves that had been in operation for a few years and the autoclave maintenance process was unknown. It is understood that the effectiveness of sterilization can be affected by factors such as lack of knowledge of sterilization protocols by the personnel in charge. Thus, it would be advisable to use chemical indicators that measure two or more parameters and periodically verify the operation of the autoclave (22,23,24).
There are standards of international and national organizations that recommend starting with proper cleaning of instruments after use. Such cleaning should be done with enzymatic detergents due to their potential to remove biological residues. Chlorhexidine is often used as a cleaning material, although it is not a detergent itself, but rather an antiseptic (22).
A study carried out by Gimenes Corrêa, et al., in Brazil (23), showed an efficiency of 100 % of the sterilization process measured with external chemical indicators. Similarly, Rodríguez, et al., in Cuba (14), found 100 % efficacy in 28 readings (14). Such findings are consistent with those of the first four autoclaves evaluated in our study. Regarding the autoclaves that were less effective, Gimenes Corrêa, et al. (23), and Tole-Acosta, et al. (24) suggest that such failures may be due to mechanical alterations of the sterilization equipment (23,24).
In relation to the type of dental office and the effectiveness of the bioburden, in this study it was observed that there was no statistical association between the variables. Santafé Viana and Izquierdo Bucheli (25) analyzed the sterilization effectiveness of dental instruments in three sterilization centers of the Central University of Ecuador’s School of Dentistry. They used biological indicators and accepted a classification of the autoclaves according to the type of office similar to that of our study. Their findings were similar to those of this investigation.
In another study, Chávez-Fermín, et al., in the Dominican Republic (26), used microbiological cultures to determine the efficacy of sterilization on endodontic and periodontal bioburdens. Of all the samples analyzed, 60 % of the files were not contaminated, nor was 69 % of the periodontal bioburdens. It has been found that, after sterilization, biological residues still persist on the surface of the instruments, which is accentuated in endodontic instruments due to the structural characteristics of their surface. The permanence of residues directly influences the obstruction of the efficient arrival of water vapor and with this the sterilization process is affected.
Regarding the variables time of maintenance and age of the autoclave, several authors recommend keeping a record of the technical maintenance carried out annually. Such processes must be monitored as they can influence the effectiveness of sterilization. However, in this study no association was found between these variables and the independent study variable (22,27).
The chemical indicators clearly indicate whether sterilization parameters such as time, temperature, pressure, and steam were met; however, they do not assess the quality of the sterilization process. Despite their limitations, the indicators are affordable and their use in routine practice is easy.
CONCLUSIONS
In the group of private practice dental offices analyzed in the city of Machala, Ecuador, most autoclaves had high efficiency. However, no statistical association was found between efficacy and the type of dental office, the maintenance time and the age of the autoclaves.
Acknowledgments
For future investigations that address the same study problem, it is recommended to use a probabilistic and representative sample, to produce generalizable results. Likewise, it is suggested to analyze the effectiveness of autoclaves in clinics in the private and public sectors.
REFERENCES
1. Cruz Quintana SM, Díaz Sjöstrom P, Arias Socarras D, Mazón Baldeon GM. Microbiota de los ecosistemas de la cavidad bucal. Rev Cubana Estomatol. 2017 Mar; 54(1): 84-99.
2. Rodríguez Uramis M, Arpajón Peña Y, Sosa Pérez AL. De la bioseguridad al control de infecciones en Estomatología. Rev Cubana Estomatol. 2014; 51(2): 224-236.
3. Hernández Castañeda AA, Aranzazu Moya GC. Características y propiedades físico-químicas de la saliva: una revisión. UstaSalud. 2012; 11(2): 102-112. https://doi.org/10.15332/us.v11i2.1123
4. Vázquez Rodríguez I, Gómez Suárez R, Estany-Gestal A, Mora Bermúdez MJ, Varela-Centelles P, Santana Mora U. Control de la infección cruzada en los laboratorios de prótesis dental de Galicia. An Sist San Navarra. 2018 abr; 41(1): 75-82. https://dx.doi.org/10.23938/assn.0169
5. Del Valle SC. Normas de bioseguridad en el consultorio odontológico. Acta Odontol Venez. 2002; 40(2): 213-216.
6. Romero MBR, Mendez PNC, Martínez NMP, et al. Comparación bacteriana de 30 piezas de alta velocidad antes y después de ser utilizadas en la Facultad de Odontología Región Veracruz. Rev ADM. 2017; 74(4): 185-188.
7. Bedoya GA. Revisión de las normas de bioseguridad en la atención odontológica, con un enfoque en VIH/SIDA. Univ Odontol. 2010; 29(62): 45-51.
8. Garrido García M, Perea Pérez B, Labajo González E. Efectividad y seguridad de los procesos de esterilización en Odontología. Acta Acad. 2013; 246: 190-198.
9. Deogade SC, Mantri SS, Saxena S, Sumathi K. Awareness and knowledge of undergraduate dental students about sterilization/disinfection methods of extracted human teeth. Ann Med Health Sci Res. 2016 Nov-Dec; 6(6): 348-355. https://dx.doi.org/10.4103/amhsr.amhsr_85_16
10. Guerra ME, Tovar V, La Corte E. Estrategias para el control de infecciones en odontología. Acta Odontol Venez. 2006; 44(1): 132-138.
11. Felzani R. Cicatrización de los tejidos con interés en cirugía bucal: revisión de la literatura. Acta Odontol Venez. 2005; 43(3): 310-318.
12. República del Ecuador, Ministerio de Salud Pública. Manual de Bioseguridad para establecimientos de salud. Quito: Dirección Nacional de Calidad, Dirección Nacional de Normalización; 2016.
13. Gimenes Corrêa E, Freire de Castilho AR, Pereira CV. Indicadores químicos e biológicos da eficácia de esterilização por autoclave ou estufa. Rev Odonto Ciênc. 2009; 24(2): 156-160.
14. Rodríguez GM, Pérez GM, Álvarez MCR. Efectividad de los integradores químicos en la esterilización por vapor a presión. Invest Medicoquir. 2018; 10(2).
15. Hernández Lomelí S, Alavez Rebollo S, García Hernández J, Flores Luna MG. Monitoreo con indicadores biológicos de rápida lectura de las autoclaves de CEYE de la Facultad de Odontología de la Universidad Tecnológica de México. Rev Odontol Mex. 2016; 20(2): 93-97.
16. Panta G, Richardson AK, Shaw IC. Effectiveness of autoclaving in sterilizing reusable medical devices in healthcare facilities. J Infect Dev Ctries. 2019 Oct 31; 13(10): 858-864. https://dx.doi.org/10.3855/jidc.11433
17. Dagher J, Sfeir C, Abdallah A, Majzoub Z. Sterilization and biologic monitoring in private dental clinics in Lebanon. J Contemp Dent Pract. 2018 Jul; 19(7): 853-861.
18. Sheth NC, Rathod YV, Shenoi PR, Shori DD, Khode RT, Khadse AP. Evaluation of new technique of sterilization using biological indicator. J Conserv Dent. 2017 Sep-Oct; 20(5): 346-350. https://dx.doi.org/10.4103/JCD.JCD_253_16
19. Argüello D, Mendoza S. El nuevo enfoque para la atención odontológica durante la pandemia de COVID-19. Revisión bibliográfica. Rev Metro Cien. 2021; 29(1): 58-64.
20. Hernández Sampieri R, Fernández Collado C, Baptista Lucio P. Metodología de la investigación. 6ta ed. México: McGraw-Hill; 2014.
21. Lara Van der Linde C, Amargos L, Streese S. Verificación de la eficacia del proceso de esterilización de la Escuela de Odontología Dr. René Puig Bentz de la Universidad Nacional Pedro Henríquez Ureña, mediante indicadores biológicos, en el periodo enero-abril, 2017. (trabajo de pregrado). Santo Domingo, RD: Universidad Nacional Pedro Henríquez Ureña; 2017.
22. Sasamoto SA, Tipple AF, Silva e Souza AC, Paiva EM, Paula e Souza CD, Pimenta FC. Evaluation of central supply units in public dental medicine colleges in Brazil. Braz J Infect Dis. 2004 Dec; 8(6): 445-453. https://dx.doi.org/10.1590/s1413-86702004000600009
23. Gimenes Corrêa E, Freire de Castilho A, Pereira CV. Indicadores químicos e biológicos da eficácia de esterilização por autoclave ou estufa. Rev Odonto Ciênc. 2009; 24(2): 156-160.
24. Tole-Acosta HD, Hernández-Roldan P, Samara-Ordoñez M. Procesos de desinfección y esterilización en centros odontológicos, revisión literaria desde el estado del arte del instrumentador quirúrgico. Rev Odontol Latinoam. 2020; 12(2): 35-45.
25. Santafé Viana JV, Izquierdo Bucheli AE. Eficacia de esterilización del instrumental odontológico en las centrales de esterilización de la Facultad de Odontología de la Universidad Central del Ecuador, mediante la utilización de indicador biológico. MetroCiencia. 2020; 28(3): 49-56. https://doi.org/10.47464/MetroCiencia/vol28/3/2020/49-56
26. Chávez-Fermín E, Domínguez-Cuevas NM, Acosta-Carrasco S, Jiménez-Hernández L, De-la-Cruz-Villa R, Grau-Grullón P, Pereyra D. Evaluación de la eficacia de la esterilización del instrumental odontológico en la Clínica de Odontología de Unibe. Rev Nac Odontol. 2013; 9(17): 35-39.
27. Portillo B, Yohanna V, Aponte F, Bertila J, Durán J. Costos de calidad en el servicio de un laboratorio clínico. Rev Técnica Fac Ingeniería Univ Zulia. 2007; 30: 309-320.
Notes
*
Original research.
Author notes
Author’s Note:a Correspondence: noheriofriob@hotmail.com; jsarmientoo@ucacue.edu.ec; estab.-sh@hotmail.es; jtorracchic@ucacue.edu.ec
Additional information
How to cite this article: Riofrio Berrú NC, Sarmiento-Ordóñez JM, Paladines Calle SE, Torracchi-Carrasco JE. Verification of the effectiveness of autoclaves through chemical indicators. Univ Odontol. 2021; 40. https://doi.org/10.11144/Javeriana.uo40.veac