INTRODUCTION
Never before as in the last two decades, hematology and, especially, hemostasis and coagulation science in a synergistic collaborative work with tissue engineering, have been so linked to regenerative dentistry. This trend leads us to think that they build a single path to become particularly important collaborative sciences, in which dentistry needs repair and regeneration and obtains from hematology the functionality of hemostasis. This is what different researchers who have obtained satisfactory results in the engineering used for tissue regeneration have wanted to verify (1-4) and that promote the repair and regeneration of tissue defects, as well as the regulation, modulation, and acceleration of inflammation and cicatrization.
The history of autogenous platelet concentrates (PC) began for science in approximately 1954 with one of the first published articles entitled “Evidence of an Antagonist to Factor VI in Platelet-Rich Human Plasma” (5). In that article, Kingsley spoke of platelet-rich plasma (PRP) referring to autologous PCs. However, other researchers claim that studies began in the 1970s with fibrin glue (6-9). The truth is that PCs have been used in dental practice for more than three decades as a regenerative tool capable of releasing supraphysiological doses of growth factors that are responsible for inducing tissue regeneration derived from autologous sources (10). With its maximum development in the last 20 years, PCs have had more supporters than detractors and more studies have shown their great advantages and contributions to tissue engineering.
The regenerative potential of platelets, the foundation of PCs, is attributed to growth factors and cytokines as they are responsible, among other processes, for increased collagen production, cell proliferation, angiogenesis, cell migration, and induction of cell differentiation (11). These benefits can be enhanced by using them in the patient's body and at the time when their physiological action is required, as is the case of periodontal defects, intraosseous defects, situations in which sinus elevation is required, alveolar preservation after extraction, and gingival recession, to name a few (12).
Due to the above, the purpose of this integrative review, the result of the compilation and analysis of existing scientific research, was to present current evidence of the benefits of PCs in the last decade and verify the efficiency of their characteristics and development for tissue engineering.
MATERIALS AND METHODS
The information search for the integrative literature review was conducted in the integrated search engine EBSCOhost, mainly in the index databases: PubMed, Scopus, Embase, and PROSPERO without language restrictions. Areas of interest were the use of plasma and platelet concentrates in dentistry, regenerative dentistry, tissue regeneration, and tissue engineering. Only studies in English and Spanish and two studies in Portuguese were included. The search was limited to the uses and investigations of PCs in the dental area in clinical studies in humans and in animal models published from 2010 to April 2021. The following keywords with their Boolean connectors were used: “platelet concentrates” OR “ platelet concentrates” AND “PRF (platelet-rich fibrin),” AND “PRP (platelet-rich plasma),” AND “tissue engineering in dentistry.” Of 3,558 titles, 71 were selected (37 reviews, 19 case reports, 9 in vitro studies, 3 animal studies, and 3 consensus). The main selection criterion was the relevance and the authors of the topic. Appendices 1-3 summarize the articles used to conducts this literature report.
RESULTS AND DISCUSSION
History
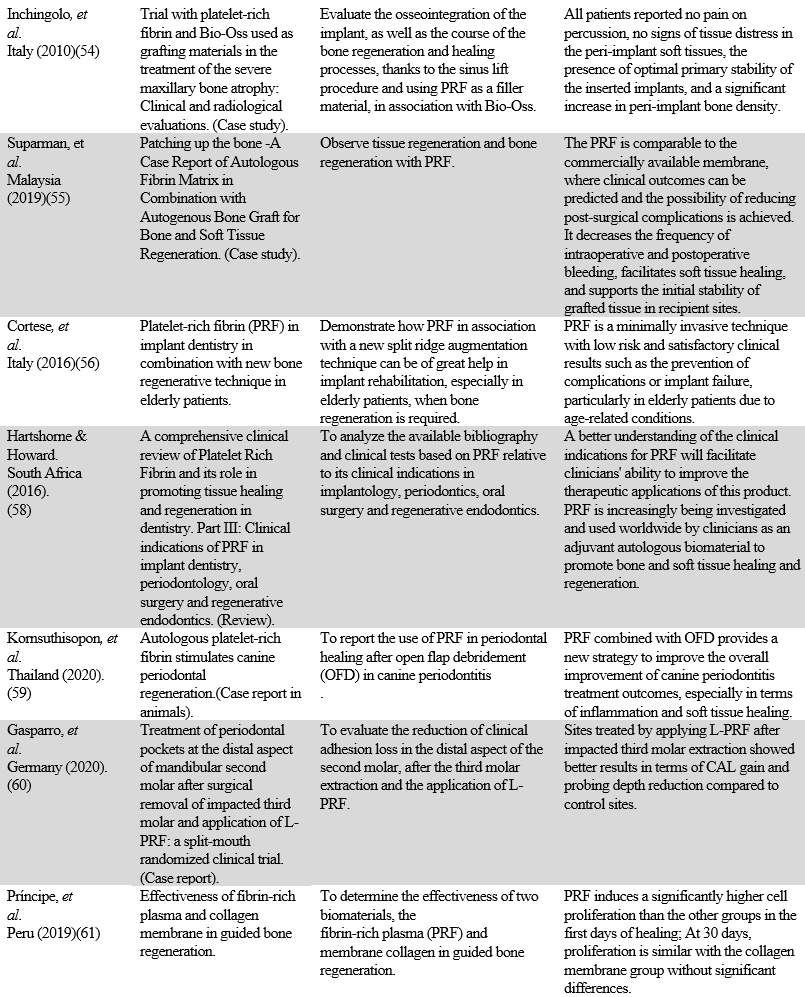
There are investigations of the use of PCs since 1940 that are attributed to Young and Medawa (9,13). Although some authors speak of the beginnings in the 1950s (14,15), most refer to the 1970s as the beginning of the application of PCs in dentistry (11,14,16-18) with the fibrin glue. The first PC protocols were described by the so-called autologous platelet-derived wound healing factors (PDWHF) and published in 1986 (13). In the 1990s, PCs and everything related to them were called PRP and it was in the new millennium that platelet-rich fibrin (PRF) and all its derivatives were created. Between 2009 and 2010, a wide variety of PCs was presented, which were called PRP to refer to the generic term used in transfusion medicine: platelet-rich plasma. This last denomination was too general for the qualification of the various products developed (19), so Dohan-Ehrenfest, et al. (19–22) classified the different platelet derivatives or PCs into four families, depending on their leukocyte content and fibrin architecture. Figure 1 shows the trajectory and evolution of PCs from 1954 to 2021.
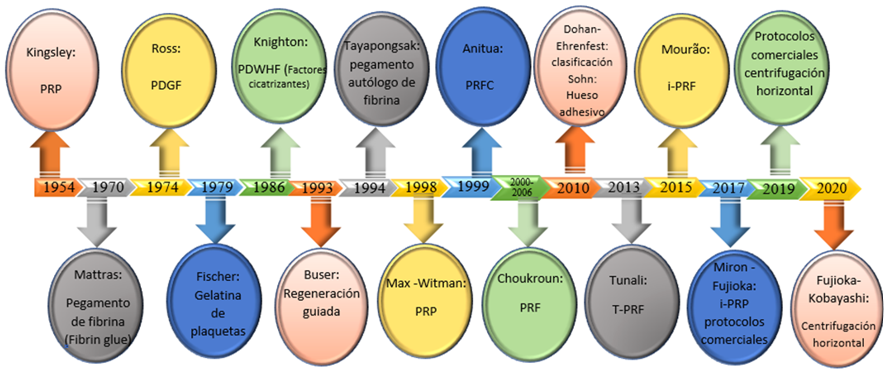 FIGURE 1
Trajectory and evolution of PCs from 1954 to 2020. Authors and creators of different technical proposals are on the list
FIGURE 1
Trajectory and evolution of PCs from 1954 to 2020. Authors and creators of different technical proposals are on the list
Source: the authors.
What Are and What Are PCs Used For in Tissue Engineering?
According to Gilberto Sammartino, autologous PCs are a concentrated suspension of growth factors and cytokines found in platelets, which function as bioactive surgical additives, and are applied locally to induce wound healing (6,11,19). PCs are increasingly used for the regeneration and reconstruction of bone and connective tissues in the periodontal and maxillofacial fields (16). Tissue engineering has produced a change in the restorative paradigm and proposes regenerative dentistry that allows the restoration of biological functions that synthetic materials cannot achieve (23,24).
The mechanism of action of PCs is the same as that of autologous healing, but with greater and repotentialized healing. The healing cascade begins with clot formation (hemostasis), which is followed by inflammation, the proliferative stage, and maturation and remodeling. PCs catalyze the proliferation of phagocytic cells that help defibrillate the injury. In turn, white blood cells and platelets release important cellular mediators such as growth factors (GFs) and cytokines that begin the healing process (2,14,18). GFs are polypeptides whose mission is to facilitate and enhance intercellular communication at the molecular level that stimulates cell migration, proliferation, and differentiation and are the main growth-regulating molecules for cells in vivo and in culture (2,25). On the other hand, these molecules, by promoting angiogenesis, mitosis, chemotaxis, and stem cell proliferation, help increase bone and soft tissue regeneration (2,18,26). Table 1 shows the most important GFs and cytokines present in PCs (2,15,16,27). Proteins such as coagulation factors and others secreted by platelets ensure initial hemostasis. After 24 to 48 hours, the proliferative phase takes over, by virtue of the presence of the inflammatory mixture of chemokines created during the inflammatory phase, to then give way to the boosted proliferation of fibroblasts, leukocytes, macrophages, and mesenchymal stem cells that begin to lay the first foundations of the new tissue. In this way, GF and other bioactive molecules form one of the essential components of the tissue engineering approach, in combination with an appropriate scaffolding and a population of stem or progenitor cells (24,25).
TABLE 1
Main Growth Factors and Cytokines Present in PCs
Source: Table translated and modified from Tomoyuki Kawase (2015) and updated from Andia and Maffulli (2018) (2,15,16,27 ).
Classification of PCs
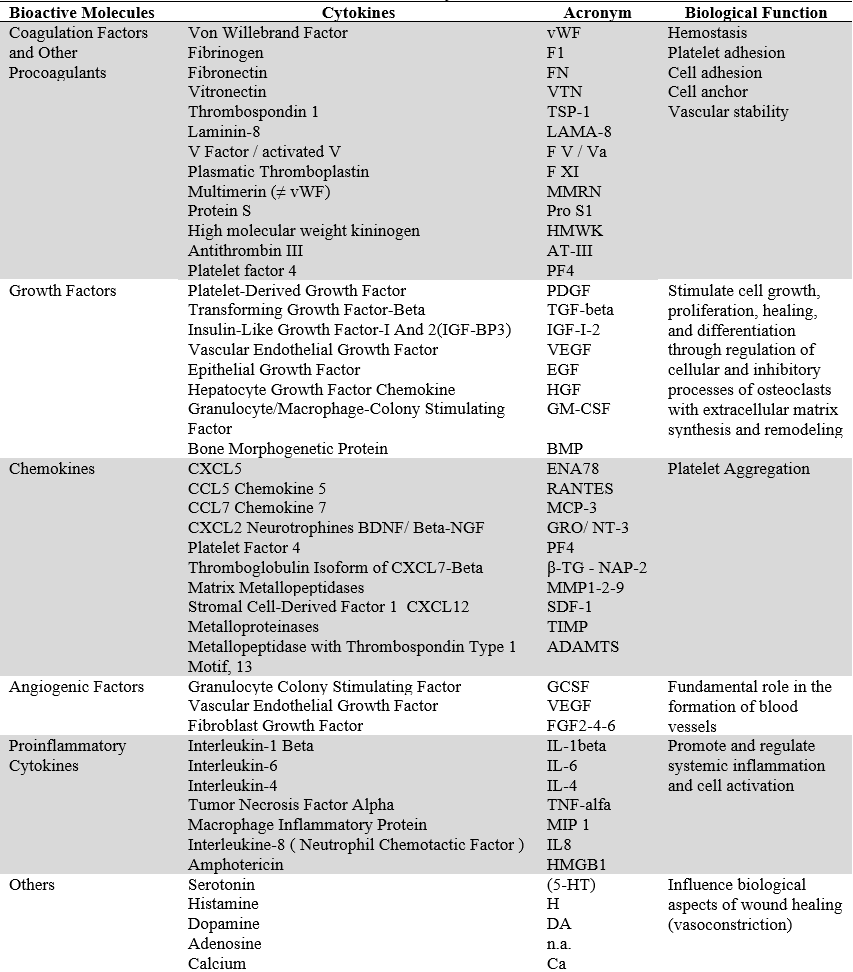
As mentioned above, the four families of platelet concentrates proposed by Dohan-Ehrenfest are pure platelet-rich plasma (P-PRP), leukocyte-platelet-rich plasma (L-PRP), pure platelet-rich fibrin (PRF), pure platelet-rich fibrin (PRF), and leukocyte- and platelet-rich fibrin (L-PRF) (15,21,28). Currently, based on this classification, there are mainly two PCs (PRF and PRP), each with its by-products (table 2).
TABLE 2
Main Characteristics of PRF y PRP (15,21,28)
Source: the authors.
Platelet-Rich Plasm (PRP)
PRP is a first-generation autologous platelet concentrate that contains a high concentration of platelets in a small volume of plasma. Its concentration ranges from 2 to 8.5 times the normal level (29). It also contains a minimal amount of natural fibrinogen and is obtained from whole blood anticoagulated with sodium citrate. It also requires calcium chloride (CaCl) and bovine thrombin to activate (initiate platelet degranulation) and initiate the coagulation process. For some authors, the presence of bovine thrombin is one of its disadvantages (21,23,30–32).
Platelets begin to actively secrete bioactive molecules within 10 minutes of clot formation. They complete the secretion of more than 95% of the pre-synthesized growth factors in one hour. Alpha granules release growth factors between one and four days after platelet activation but sustain their stimulation of the proliferative phase for approximately 10 days after release. It has been shown that, in addition to growth factors, the granular tissue of PRP-treated wounds contains intra- and extra-platelet components that also contribute to tissue regeneration. When the direct influence of platelets begins to decrease, the macrophages that arrive through the vascular stream are stimulated by the platelets and assume responsibility for the regulation of healing by secreting their own factors (14,18,28,29,34).
The group of Anitua, et al. (34) have been the great promoters of the PRP preparation. They established the simplest and most popular technique for its preparation (2). Certainly, it should be considered that modifications in the protocol can change the platelet concentration factor (30) (Table 2).
Uses of PRP
There are numerous applications of PRP, not only in regenerative dentistry but also in maxillofacial surgery, ulcer treatment, spinal surgery, orthopedic surgery, and aesthetic medicine (13,16,22).
Periodontics
PRP is used as an adjuvant to regenerative therapy (22,35). In this sense, Okuda-Kawase, et al. (36) show an increase in DNA synthesis during the periodontal regeneration process in patients treated with PRP. When evaluating the mitogenic activity through the incorporation of 5-bromodeoxyuridine, these researchers found a significantly higher cell count in the periodontium of patients treated with PRP in relation to a control group (36).
Implantology
When preparing the maxillary bone for implant placement, there is greater buccolingual/palatal bone width, greater bone density, and faster tissue coverage in the alveoli where PRP is used (35). Another comparative study showed that the addition of PRP with beta-tricalcium phosphate (beta-TCP) results in less bone loss around implants (38). These findings reaffirm the multiple publications by Anitua (34) in which bone regeneration, osseointegration, and soft tissue gain are confirmed when using PRP alone or combined with other substances such as bone substitutes. The properties of PRP have also been evidenced in the management of gingival recession defects, alone or in combination with different guided bone regeneration techniques and biomaterials (2).
Maxillofacial and Reconstructive Surgery
PRP is used to accelerate bone healing and maturation which improves alveolar healing (17,22,23). PRP improves bone regeneration and leads to platelets acting as local regulators of the healing process by increasing the microcirculation of the gingival mucosa surrounding the wound (35)
Maxillary Floor Elevation
An example is a clinical case published in Mexico (29) that was evaluated for seven years, in which maxillary sinus floor elevation was performed using plasma rich in growth factors (GFRP), absorbable hydroxyapatite, and bone allograft as graft materials, and two implants were placed simultaneously. That study concluded that using GFRP makes it possible to simplify the technique by compacting the subantral graft, in sinus elevation, which improves consistency, facilitates handling, and increases the amount of graft. In that study, the radiological evaluation showed optimal results, because, in addition to reducing the total treatment time, it was effective in improving soft tissue healing and as was the purpose, increasing bone volume in the posterior maxillary area to implant placement. Other reports in the literature show a high success rate when using this technique and make it a predictable, effective, and safe procedure (29).
Healing of Mucous Tissue After Extraction
A study by Bonilla, et al. (33) shows the effective healing action on mucosal tissue after third molar extraction by comparing the healing action between mucosal tissue treated with PRP and control tissue. When performing the extraction and observing the improvement in the postoperative period of the patients, they concluded that using PRP, due to the high content of growth factors, helps to accelerate the healing process, prevents the tissues from becoming inflamed, and reduces postoperative pain (33). Acosta, et al. (38) also confirmed these findings, in an in vitro study that evaluated the proliferation and cell viability of fibroblasts and osteoblasts of the periodontal ligament stimulated with PRP. They found statistically significant differences when PRP was used in cell cultures. Additional evidence shows that the rate of alveolar osteitis is lower in bone sites treated with GFRP (18).
Antimicrobial Action
CieVlik-Bielecka, et al. (39), using the Kirby-Bauer disk diffusion method, found that L-PRP inhibited the growth of Staphylococcus aureus and was active against Enterococcus faecalis and Pseudomonas aeruginosa. They also found that a high concentration of thrombin, which, as an activator, increases the potency of the antimicrobial action of L-PRP (39). Hartshorne and Gluckman (40) explain the antimicrobial activity to the diverse populations of leukocytes and stem cells present in PRP. On the other hand, PRP can provide early protection against bacterial contamination (16) during surgical procedures, but it is cautioned that, although the antibacterial action of PRP is favorable, it should not be overestimated or considered comparable in efficacy with antibiotic therapy, according to Kawase (16). After reviewing various studies, PRP has also inhibited the growth of bacteria such as Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Klebsiella sp.
Platelet Rich Fibrin (FRP) or Fibrin Rich Plasma (FRP)
PRF is a fibrin matrix with platelets, cytokines, and GF, which acts as a biodegradable structure not requiring additives. The enhanced fibrin structure protects developing tissues and controls the gradual release of GF as the membrane degrades (2). Actually, PRF is a second-generation technology (4,16,17,21,25,41-43) based on PRP but, by preserving growth factors in a three-dimensional matrix, it can exert its effects days or weeks after surgery (26). PRF also contains fibronectin and vitronectin that promote angiogenesis. Within minutes after phlebotomy and contact with the collection tube material, the absence of anticoagulant allows most of the platelets contained in the collected blood to be activated to trigger the coagulation cascade. Fibrinogen is concentrated until the effect of thrombin circulation transforms it into a three-dimensional network capable of trapping molecules and cells (fibrin network) that also has the characteristic of acting as a barrier membrane in bone regeneration procedures and guided tissue regeneration, that is, GBR and GTR (6,24,25,41,43).
Specifically, PRF is a three-dimensional structure composed of fine, flexible, mature, and dense polymerized fibrin strands. In the structure, the red blood cell zone is trapped in a relatively immature fibrin network (17). At the junction of the red corpuscular zone (RBC) and the fibrin body, lymphocytes with an irregular surface are observed microscopically along with numerous platelet aggregates. Groups of large and dense platelets are observed in the area of the buffy coat, indicating that they are in a state of aggregation and coagulation. In the remainder of the fibrin body, a thick mature fibrin strand is seen running parallel to each other. Platelets are concentrated in the first millimeter after the red part and steadily decrease with increasing distance from the red end (17). Figure 2 shows the structural characteristics and distribution of the PRF components.
The histological analysis performed by Gutiérrez, et al. (44) showed that there is no homogeneous platelet distribution throughout the FRP membrane. In the upper part, which is called platelet poor fibrin (PPF), there is a lower platelet concentration than in the lower BC (white blood cell layer) area. In the RBC, layers of polymorphonuclear cells and erythrocytes are observed. Regarding fibrin, it is homogeneous in most of the FRP, except in the upper zone that is a bit loose. In the same study, scanning electron microscopy (SEM) analyses confirmed that the structure of the fibrin network and the cellular content are different in the three zones of the membrane, the concentration of platelets is higher in zone BC, and it is diminished in the PPF zone. Likewise, fibrin in zone BC is denser (29,33,36,44).
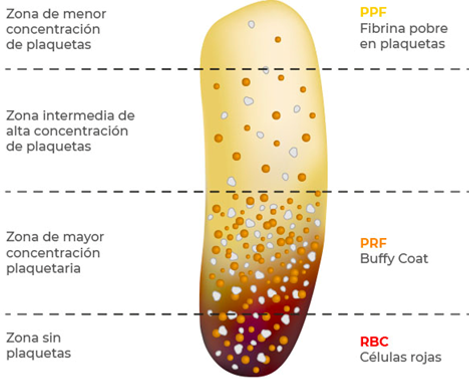 FIGURE 2
Structural Characteristics and Distribution of PRF Components
FIGURE 2
Structural Characteristics and Distribution of PRF Components
Figure adapted by the authors (29,33,36,44)
PRF provides a temporary matrix for the injured tissue until remodeling is complete. It is important to note that the preparation and formation of the fibrin network must be tightly controlled as it influences the final properties of the product (24,45-47).
In 2020, Thanasrisuebwong, et al. (46) evidenced the physical and biological properties of i-PRF compared to PRP. They used techniques such as SEM and ELISA and analyzed viscoelastic properties using the rotational thromboelastometry technique (ROTEM®) (46). To demonstrate the effects of C-PRF on angiogenesis, tissue growth studies in chicken eggs have counted the number of vessels formed during the incubation period and have compared it to baseline (day 0). Researchers found the formation of new capillary blood vessels, cross-linked with each other to generate networks, as well as the C-PRF content containing PDGF isoforms at higher levels, which significantly stimulated cell proliferation and neovascularization (36).
Apoorv Goel, et al. (43), also in 2020, concluded in a study that PRF membranes in combination with their exudates result in a high cell-proliferation rate. This basically translates into great therapeutic and wound healing potential that is not affected by a patient's periodontal condition.
PRF is used in combination with bone grafts (bovine porous bone mineral), nanocrystalline hydroxyapatite, and freeze-dried demineralized bone allograft (DFDBA), or pharmacologic agents, such as metformin gel. PRF has been found to be more effective in improving clinical and radiographic parameters in patients whose grafts were treated with PRF (1,6,25). When compared to PRP, PRF preparations tend to have a higher leukocyte count, due to improvements in preparation techniques (14,25).
Since its creation in 2001 by Choukroun, et al. (11,17,21,28,31,48), several modifications have been made to the conventional protocol to prepare PRF: Advanced PRF (A-PRF®) which is PRF collected in glass tubes and centrifuged at a slower speed; Injectable PRF (i-PRF®), which has the minimum centrifugation time and its application or injection is immediate; Lysed PRF (PRF LySATE®), which is obtained from PRF that is incubated at 37 °C in a humidified atmosphere of 5% CO2 / 95% air; and Titanium PRF (T-PRF®), which comes from the PRF that is collected in medical-grade titanium tubing. All variations of PRF use different centrifugation conditions, with the basic structural design, and use different collect tube materials (11,49).
Over the last decade, PRF has gained tremendous momentum for being used in a wide variety of dental and medical procedures (6,10,17,50,51). The ability of PRF to stimulate regeneration in a wide range of tissues has been repeatedly found. PRF has been shown to rapidly stimulate tissue healing by significantly increasing the recruitment and proliferation of a variety of cells, including endothelial cells, gingival fibroblasts, chondrocytes, and osteoblasts. In this way, it promotes tissue repair and angiogenesis at the injury site (4,16,17,20–22,25,41,51).
Main Uses of PRF in Clinical Dentistry as a Membrane
In surgery, PRF is used as a resorbable membrane for guided bone regeneration (GBR) that includes alveolar ridge augmentation (11,37,51) and prevents undesirable cell migration into bone defects. PRF also provides a space for osteogenic and angiogenic cell adhesion, allowing the underlying blood clot to mineralize (16,52). This membrane also protects open wounds from the oral environment when sutures cannot bridge the mucosal margins and has shown favorable clinical results in the treatment of intra-bone periodontal defects (11,51-53).
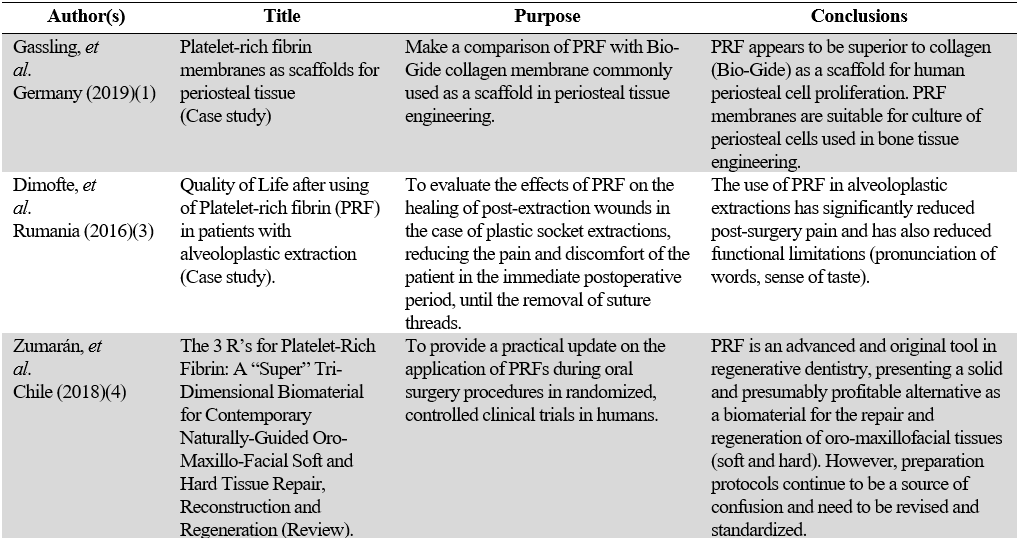
In 2010, Gassling, et al. (1) showed the superiority of PRF over collagen (Bio-Gides®) as a scaffold for periosteal cell proliferation in periosteal tissue engineering. Other clinical studies with promising results used PRF membranes as sole graft material to seek augmentation of the maxillary sinus floor (11). Since PRF does not require the use of other biological materials to cover an exposed flap, it offers the additional advantage of not creating a risk of infection, if exposed to the oral cavity. It has been proven that, within a three-month healing period, the fibrinous matrix transforms into new tissue by regenerating bone in the soft-tissue-lined cavity. Another randomized clinical trial, reported in the systematic review by Miron, et al. (53), published in 2017, showed that the exclusive use of PRF (studies by Inchingolo, et al. [54]), before implant placement, could minimize dimensional changes after extraction and improve osteogenesis (53). In all the cases of said study, the authors observed a satisfactory implant-prosthetic rehabilitation, according to the Albrektsson criteria (54). By applying these criteria, a success rate of 85% at 5 years of observation and 80% at 10 years of observation is expected to classify the implant at the minimum levels of success expected.
The concept of socket preservation was implemented to minimize bone resorption after tooth extraction and to preserve alveolar bone using PRF. A clinical study with 117 patients, after dental extraction, showed the benefit of PRF in preventing alveolar osteitis and improving hemostasis (15). A PRF membrane can also be used to improve wound healing in immunocompromised patients and as an adjuvant in patients on anticoagulant therapy (11).
PRF in Oral and Maxillofacial Surgery and Dental Implantology
In most clinical studies in implantology, the use of PRF focuses on improving clinical outcomes, for example, in sinus lifts, using PRF as the sole grafting material simultaneously with implant placement, or using a combination of PRF and bone allograft (FDBA) prior to implant placement. Other focus areas of clinical research are implantology with the use of PRF in preservation of the alveolar ridge, healing of the tissues around the implant, and reduction of mobility, due to the rapid healing of the tissues surrounding the implant and efficiency of osteogenesis, which improves its stability. In vitro studies have shown gene expression (PRF-induced) of early and late markers of osteogenesis, bone marrow stimulation, and soft tissue healing (13,27,40,49,55,56).
PRF can also be used as a filling material, especially in multiple extractions, to preserve the height of the alveolar ridge. In such cases, PRF acts as a super blood clot for neovascularization and acceleration of tissue regeneration (4,53). This fact was also shown by Cortese, et al. (56), in 10 elderly patients, by promoting neo-angiogenesis. They concluded that the most relevant advantages of using PRF are the healing and regenerative properties of the bone and its complete resorption after surgery, which leads to avoiding a second surgery, an especially important fact in patients with advanced age (56,57).
Hartshorne and Gluckman (40) showed in extensive studies, results of improvement in the clinical performance of paranasal sinus lifts (11). PRF in combination with bone graft materials has been used in several direct and indirect sinus lift techniques, including bone graft sinus floor lift, osteotome-mediated sinus floor lift, and osteotome-mediated sinus floor lift, minimally invasive antral membrane balloon techniques (11). Other studies in which PRF was used alone as a graft material for sinus lift led to the conclusion that PRF significantly promoted bone healing with particularly good bone gains, between the sinus floor and the upper part of the alveolar crest, at 6 months, 1 year, and 6 years, preservation of the implant was obtained (53,58).
In cases of wide cavities and lesions where primary closure is difficult, a PRF membrane can be used as a lining and protection membrane that promotes re-epithelialization of the site and accelerates the union of the wound margins. The resistance and elasticity of the PRF membrane make it easy to suture. As a membrane for GBR, the dense matrix architecture of PRF covers, protects, and stabilizes the bone graft material and the operative site. In addition, PRF can be used to treat peri-implant bone defects and to regenerate bone and soft tissue in immediate implants (11).
PRF in Periodontics
Currently, most clinical studies in periodontics focus on improving clinical outcomes with treatment of intra-osseous periodontal pockets, furcation defects, gingival recession defects, healing of connective tissue graft sites in the palate (13,58), oral lesions such as ulcer or open sore, and periapical lesions (29). A case study by Hernández Tejeda in Mexico (29), with a potential root coverage approach (11,29), reported the use of a combination of PRF gel, hydroxyapatite graft, and membrane-guided tissue regeneration to treat an open sore injury. PRF can be used to cover localized gingival recession in mandibular anterior teeth by the combined technique of laterally placed flap and PRF membrane, to stimulate osteogenic differentiation of dental pulp cells (11). The relevance of PRF in periodontal regeneration is attributed mainly to platelet-derived growth factor (PDGF) and transforming growth factor (TGF). For its part, the production of osteoprotegerin (OPG) causes the proliferation of osteoblasts (11,35,37).
In 2020, a study with 23 dogs showed evidence of the use of PRF in periodontal healing after open flap debridement (OFD) in canine periodontitis. In the OFD plus PRF group, a considerably decreased inflammatory score was observed, when compared to the OFD group alone or the control group. Also, collagen accumulation improved in the OFD + PRF group at a later time, when compared to the baseline. The application of PRF also significantly reduced the expression of inflammatory cytokines (TNFA and IL1B) and promoted the expression of genes related to the production of collagen (COL1A1, COL3A1, and TIMP1) and growth factors (PDGFB, TGFB1, and VEGFA) (59). .
As has been shown, the mandibular third molar can cause periodontal defects in the distal root of the second molar. Gasparro, et al. (60) conducted a split-mouth randomized clinical trial to treat periodontal pockets in the distal mandibular second molar after surgical extraction of the impacted third molar with application of L-PRF®. After six months, sites treated with L-PRF® showed better results in terms of CAL clinical attachment level gain and depth reduction when compared to sites that did not have L-PRF® applied (60). In a panel discussion in 2020, while celebrating 20 years of continued use of biologic products in medicine and reviewing existing evidence, it was concluded that periodontal regeneration must show evidence of new bone, cementum, and periodontal ligament on the surface of previously diseased tooth root. This should be verified histologically because clinical findings can be difficult to identify and are not very objective (57). In different periodontal procedures, the elastic consistency of the PRF membrane also allows a hole to be drilled in the membrane to hang a healing abutment prior to flap suture (40).
PRF in Intrabony Defect Regeneration
Randomized clinical trials have shown that the use of PRF leads to periodontal repair, with statistically confirmed superior results for intrabony defects than when PRF is not used. When using PRF, repotentiation and increased effective performance of biomaterials such as bone grafts or collagen barrier membranes are shown, in addition to periodontal regeneration of intrabony defects (53,61). A case described by Miron, et al. (53) showed results of the potential for tissue repair using PRF in furcation defects. It also showed that PRF-enriched palatal dressing significantly accelerated the healing of a palatal wound.
Recently, Miron, et al. (51) performed a systematic review and meta-analysis to conclude that open flap debridement in conjunction with PRF showed significantly higher values for probing depth, clinical periodontal attachment level, and bone formation gain. Such findings were confirmed with radiographic recording that showed the regenerative properties of PRF. They concluded that the combination of PRF with bone grafts or small biomolecules may offer important clinical advantages.
PRF to Cover Roots with Gingival Recessions
PRF has also been widely used as a bioactive matrix in numerous studies for the coverage of roots with gingival recessions. The use of coronally advanced flap (CAF) was compared with CAF + PRF. PRF was found to induce a significant increase in root coverage (4,6,18,41,53).
PRF in Endodontics
The combination of PRF membrane, as a matrix, and mineral trioxide, in apexification procedures, shows to be an effective alternative to create artificial barriers at the end of the root and induce faster periapical healing, in cases of large periapical lesions, by using a membrane can prevent extrusion. PRF has also been used to fill bone defects after periapical surgeries, such as root-end resection (resorption). PRF is an ideal scaffold for the revascularization of immature permanent teeth with necrotic pulps (11,13,63). These procedures, according to Hartshorne and Gluckman (40,58), are biologically designed to restore the function of a damaged pulp by stimulating dental pulp stem cells and progenitor cells present in the root canal under conditions that are favorable for their differentiation.
Sohn’s Sticky Bone
Sohn's sticky bone is a combination of I-PRF and graft particles, which allows the bone graft to be easily shaped and manipulated, to obtain a good consistency and to reduce leaching of the graft, since it is firmly encapsulated in the fibrin matrix (17.37). Sticky bone has the advantage of releasing growth factors at the recipient site, which would otherwise be missing from a regular bone graft. It has the potential of converting any osteoconductive graft into an osteoprotective one (due to the presence of platelets and growth factors), which would result in faster and more efficient bone formation (17). Sticky bone retains much of its shape during the healing process, with success primarily in alveolar crest and sinus augmentation (14,17,37,55).
Implants placed in cavities with CP are more stable and experience less resorption than implants placed in untreated bone. The greatest reported advantage of combining PRF with a bone graft material is decreased overall healing time and better handling of the graft material (53).
PRF Antimicrobial Effects
A split-mouth design study showed that, by placing PRF in third molar extraction sockets, an almost 10-fold decrease in osteomyelitis infections can be expected (53), due to the increase in white blood cells. The different types of leukocytes are concentrated in the fibrin matrix, of these, the monocytes infiltrate the injury and differentiate into macrophages, which produce collagenases and provide antimicrobial properties to "clean" the wound (40). In addition, macrophages have been shown to be key elements in osteogenesis, both during bone modeling and remodeling, and in association with bone biomaterials (16). Jasmin, et al. (63) evaluated the in vitro antimicrobial effects of i-PRF against pathogenic oral biofilm producing staphylococcal bacteria isolated from patients with dental and oral abscesses. In that study, the antibacterial activity of i-PRF was determined using the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) techniques. i-PRF showed bactericidal activity against both biofilm-producing and non-biofilm-producing bacteria and it was concluded that i-PRF could be a potential antimicrobial peptide to combat postoperative infections caused by biofilm-producing staphylococcal microorganisms (63).
Another study by Khorshidi, et al. (64) compared the antimicrobial, mechanical, and histological characteristics of the L-PRF membrane before and after the addition of silver nanoparticles (SNPs). Results showed a marked improvement in the mechanical properties and antibacterial activity of L-PRF after the addition of such particles. Only growth of Klebsiella pneumoniae, and no other bacteria, was found at 24 hours (65). For its part, in the clinical trial by Daugela, et al. (65) cited Tang’s findings in the article, "Antimicrobial Peptides of Human Platelets," shows how activated platelets and activated leukocytes release antimicrobial peptides, among which are platelet factor 4, RANTES, connective tissue activating peptide III, platelet basic protein, thymosin beta-4, fibrinopeptide B and fibrinopeptide A, which together with fibrinogen degradation products, are bioactive substances that have been shown to be potent antimicrobials against Escherichia coli and Staphylococcus aureus (66).
PRF in Pain Treatment
Several studies mention the additional advantage of PRF to manage pain and reduce postoperative discomfort in different procedures (3,13,24,27,33,39,52). In this sense, two more studies conducted by Daugela, et al. (65) and Canellas, et al. (67) evaluated the influence of L-PRF and its impacts on wound healing after extraction of the mandibular third molar (MTM), in addition to the benefits obtained in the patient's postoperative discomfort and the incidence of alveolar osteitis. In all cases, a decrease in pain was reported in the pain scale scores (VAS) (65,67). The use of PRF in alveoloplastic extractions has significantly reduced post-surgery pain and caused fewer functional limitations (e.g., pronunciation of words, sense of taste), improving the patients’ quality of life after use. Although these are subjective aspects, say the authors, the decrease in pain intensity and post-surgery analgesic consumption were also described by other studies (3) that were showing a healing time that was considerably shorter when using the PRF application after dental extractions (3).
PRP Compared to PRF
Due to availability and profitability, PRF has obtained more relevance, regarding uses in dentistry, in relation to its predecessor, PRP (50). PRF membranes have no contraindications; they can be used in all types of patients, especially in patients with systemic conditions whose healing is compromised (e.g., diabetics and smokers) and PRP cannot be used in anticoagulated patients (50). Several studies claim that PRF accelerates tissue growth even more, compared to PRP (14,16,25,27,38) (Table 3).
He, et al. (68) compared the effect of PRF and PRP on cell proliferation and osteoblast differentiation in an animal model with rats and quantified the values of PDGF-AB and TGF-Beta1. The researchers found that PRP releases large amounts of these factors during the first day, while PRF releases TGF-Beta1 at day 14 and PDGF-AB at day 7, corroborated by Sunil, et al. (18), and Miron, et al. (42) who reviewed several studies referring to it.
TABLE 3
PRF Compared to PRP
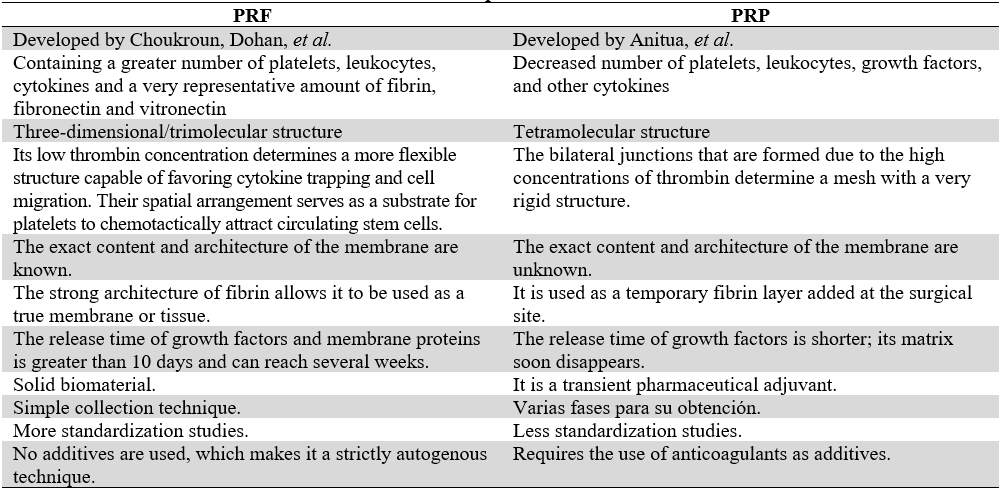
Source: the authors
The fact that there are some differences between these platelet concentrates does not prevent them from being used jointly and complementarily, as shown by Tsai, et al. (68). Those researchers in a study with induced wounds in pigs used mixtures of both PRP and i-PRF to make a platelet patch (PP) and promote wound repair and regeneration. Results indicated that all wounds showed significant size reduction (69).
Commercial Systems of CP Preparations
There are registered commercial techniques to prepare platelet concentrates with predefined times and fixed regulations for centrifugation. There are also CP already prepared industrially with trademark registration and patenting. Table 4 shows the best known commercial products and systems (10,14,16,18,19,27,45,50,58,70).
TABLE 4
Techniques and Trademarks of CP Preparations (10,14,16,18,19,27,445,50,58,70)
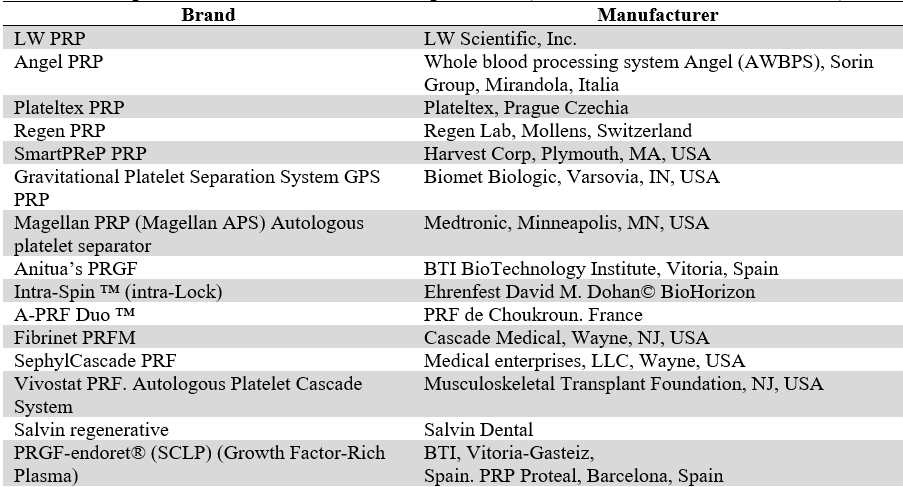
Source: the authors.
In 2014, an evaluation of 7 commercial PRP systems was performed in which platelets, white blood cells (WBC), red blood cells (RBC), and growth factors were quantified. The study concluded that differences in the purification method used to produce PRP can lead to notable differences. Other differences occurred in the amount of whole blood required, final yield, cellular components, and growth factor concentrations (70). Knowledge of these differences can lead to commercial kits or systems being used depending on the required clinical purposes and making better decisions.
A comparative study of commercial PRF systems, conducted by Dohan Ehrenfest, et al. (45), with L-PRF (Intra-Spin, Intra-Lock), A-PRF 12 (Advanced PRF, Process), LW-UPD8 (LW Scientific) and Salvin1310 (Salvin Dental). The study quantified growth factors (by ELISA technique) and found that PRF, when analyzed by light microscopy, shows relatively similar characteristics in all L-PRF centrifugation kits. The authors state that there are differences in the macroscopic structure, which led them to conclude that the characteristics of the centrifuge and the centrifugation protocols have a significant impact on cells, growth factors, and L-PRF architecture (46).
Limitations of Use and Implementation of CPs
Certainly, there are no drawbacks that discourage the use of PC, but due to the lack of diffusion that studies deserve, detractors of PC emphasize their limitations. These limitations can be attributed to flaws in the preparation protocols, due to lack of standardization, of which the variability of the results between one patient and another depends in part on. These variabilities ultimately make it difficult to predict and reproduce results.
It is necessary to treat PC as a surgical biomaterial in which asepsis and rigor are controlled in all manufacturing steps. If these requirements are not conducted properly, they could become a critical aspect for the results and efficacy of clinical and surgical procedures (28,44,62,63).
Another limitation occurs when the amount of blood collected is not sufficient, when large amounts of PC are necessary, since, as they are autologous products, their quantity is limited. It must be considered that PRF membranes should be used within a short time after preparation, since they tend to shrink as a result of dehydration (11). Currently, there is little information on the restrictions that exist for the use of PC, in patients with coagulation disorders or who use drugs that affect blood coagulation (i.e., heparin, warfarin, or platelet inhibitors) (50).
CONCLUSIONS
CP, especially PRF, have become a tissue-targeted solution in surgical situations where protection, stimulation of healing, and regeneration are critical, and where the prognosis for tissue repair is poor, or is potentially compromised. These facts make CP a novel tool for tissue engineering (2,50). However, further research and standardization of procedures is needed to support the beneficial effects of these biologics.
Platelet concentration levels in PC range from less than 2 to 8.5 times the normal level, obtaining the same regenerative benefit in each case (17). It can be said that CP can function as an initiator and enhancer of autologous healing, having synergistic effects as regenerative material, serving in dentistry as a support for conventional surgical processes that improve results in clinical and radiographic parameters. It is evident that the use of CP as a biomaterial reduces postoperative incompatibility (due to its regenerative, antimicrobial, and pain-reducing activity).
The basic structure of the matrix and cellular composition of the different PCs is the same. What changes is the physical presentation and, in some cases, the additives and added particles.
The clinical effects of CP depend on the intrinsic, versatile, and adaptive characteristics of the patient's blood and on the standardization and validation of the protocols used for its preparation.
Those derived from recombinant sources (manufactured in mammalian cells or bacteria) introduce high supraphysiological concentrations in the regeneration zones and are developed industrially. They are approved by the US Food and Drug Administration and are marketed at high prices. The different CPs generate growth factors in a natural, dosed form, and are manufactured exclusively from autologous sources. They allow to reduce costs considerably and lead to a comparable benefit in the desired clinical results.
References
1. Gassling V, Douglas T, Warnke PH, Açil Y, Wiltfang J, Becker ST. Platelet-rich fibrin membranes as scaffolds for periosteal tissue engineering. Clin Oral Implants Res. 2010 May; 21(5):543-549. https://doi.org/10.1111/j.1600-0501.2009.01900.x
2. Rodríguez Flores J, Palomar Gallego MA, Torres García-Denche J. Plasma rico en plaquetas: Fundamentos biológicos y aplicaciones en cirugía maxilofacial y estética facial. Rev Esp Cir Oral y Maxilofac. 2012; 34(1): 8-17. https://doi.org/10.1016/j.maxilo.2011.10.007
3. Dimofte M, Choukroun J, Moscalu M, Popescu E. Quality of life after using of platelet rich fibrin (PRF) in patients with alveoloplastic extraction. Rev Cercet si Interv Soc. 2016; 55: 31-46.
4. Zumarán CC, Parra MV, Olate SA, Fernández EG, Muñoz FT, Haidar ZS. The 3 R's for Platelet-Rich Fibrin: A "Super" Tri-dimensional biomaterial for contemporary naturally-guided oro-maxillo-facial soft and hard tissue repair, reconstruction and regeneration. Materials (Basel). 2018 Jul 26; 11(8): 1293. https://doi.org/10.3390/ma11081293
5. Kingsley CS. Blood coagulation: evidence of an antagonist to factor VI in platelet-rich human plasma. Nature. 1954 Apr 17; 173(4407): 723-4. https://doi.org/10.1038/173723a0
6. Verma UP, Yadav RK, Dixit M, Gupta A. Platelet-rich Fibrin: A Paradigm in Periodontal Therapy - A Systematic Review. J Int Soc Prev Community Dent. 2017 Sep-Oct; 7(5): 227-233. https://doi.org/10.4103/jispcd.JISPCD_429_16
7. Borie E, Oliví DG, Orsi IA, Garlet K, Weber B, Beltrán V, Fuentes R. Platelet-rich fibrin application in dentistry: a literature review. Int J Clin Exp Med. 2015 May 15; 8(5): 7922-7929.
8. Castro AB, Meschi N, Temmerman A, Pinto N, Lambrechts P, Teughels W, Quirynen M. Regenerative potential of leucocyte- and platelet-rich fibrin. Part A: intra-bony defects, furcation defects and periodontal plastic surgery. A systematic review and meta-analysis. J Clin Periodontol. 2017 Jan; 44(1): 67-82. https://doi.org/10.1111/jcpe.12643.
9. Matras H. Die Wirkungen verschiedener Fibrinpräparate auf Kontinuitätstrennungen der Rattenhaut [Effect of various fibrin preparations on reimplantations in the rat skin]. Osterr Z Stomatol. 1970 Sep; 67(9): 338-59. German.
10. Miron RJ, Fujioka-Kobayashi M, Hernandez M, Kandalam U, Zhang Y, Ghanaati S, Choukroun J. Injectable platelet rich fibrin (i-PRF): opportunities in regenerative dentistry? Clin Oral Investig. 2017 Nov; 21(8): 2619-2627. https://doi.org/10.1007/s00784-017-2063-9.
11. Sanjeevi J, Santhosh-Kumar MP. [Platelet-rich fibrin in dentistry]. Drug Invent Today. 2019;12(1): 158-161.
12. Wang Z, Mudalal M, Sun Y, Liu Y, Wang J, Wang Y, et al. The Effects of Leukocyte-Platelet Rich Fibrin (L-PRF) on suppression of the expressions of the pro-inflammatory cytokines, and proliferation of schwann cell, and neurotrophic factors. Sci Rep. 2020; 10(1): 1-11. http://dx.doi.org/10.1038/s41598-020-59319-2
13. Ghanaati S, Herrera-Vizcaino C, Al-Maawi S, Lorenz J, Miron RJ, Nelson K, Schwarz F, Choukroun J, Sader R. Fifteen years of platelet rich fibrin in dentistry and oromaxillofacial surgery: how high is the level of scientific evidence? J Oral Implantol. 2018 Dec; 44(6): 471-492. http://dx.doi.org/10.1563/aaid-joi-D-17-00179
14. Fan Y, Perez K, Dym H. Clinical Uses of platelet-rich fibrin in oral and maxillofacial surgery. Dent Clin North Am. 2020 Apr; 64(2): 291-303. http://dx.doi.org/10.1016/j.cden.2019.12.012
15. Dimova C, Evrosimovska B, Popovska M. El uso de materiales de injerto y plasma rico en plaquetas en cirugía oral. DDF 2017; 376: 39-53. https://doi.org/10.4028/www.scientific.net/ddf.376.39.
16. Kawase T. Platelet-rich plasma and its derivatives as promising bioactive materials for regenerative medicine: basic principles and concepts underlying recent advances. Odontology. 2015 May; 103(2): 126-135. https://doi.org/10.1007/s10266-015-0209-2
17. Shah R, M G T, Thomas R, Mehta DS. An Update on the Protocols and Biologic Actions of Platelet Rich Fibrin in Dentistry. Eur J Prosthodont Restor Dent. 2017 Jun; 25(2): 64-72. https://doi.org/10.1922/EJPRD_01690Shah09
18. Sunil M. Platelet-Rich Plasma and Platelet-Rich Fibrin in Periodontal Regeneration: A Review. Asian J Pharm Clin Res. 2017; 7(10): 1-5.
19. Dohan Ehrenfest DM, Bielecki T, Del Corso M, Inchingolo F, Sammartino G. Shedding light in the controversial terminology for platelet-rich products: platelet-rich plasma (PRP), platelet-rich fibrin (PRF), platelet-leukocyte gel (PLG), preparation rich in growth factors (PRGF), classification and commercialism. J Biomed Mater Res A. 2010 Dec 15; 95(4): 1280-1282. https://doi.org/10.1002/jbm.a.32894
20. Dohan Ehrenfest DM, Andia I, Zumstein MA, Zhang CQ, Pinto NR, Bielecki T. Classification of platelet concentrates (Platelet-Rich Plasma-PRP, Platelet-Rich Fibrin-PRF) for topical and infiltrative use in orthopedic and sports medicine: current consensus, clinical implications and perspectives. Muscles Ligaments Tendons J. 2014 May 8; 4(1): 3-9
21. Dohan Ehrenfest DM, Bielecki T, Mishra A, Borzini P, Inchingolo F, Sammartino G, Rasmusson L, Everts PA. In search of a consensus terminology in the field of platelet concentrates for surgical use: platelet-rich plasma (PRP), platelet-rich fibrin (PRF), fibrin gel polymerization and leukocytes. Curr Pharm Biotechnol. 2012 Jun; 13(7): 1131-1137. https://doi.org/10.2174/138920112800624328
22. Xu J, Gou L, Zhang P, Li H, Qiu S. Platelet-rich plasma and regenerative dentistry. Aust Dent J. 2020 Jun; 65(2): 131-142. https://doi.org/10.1111/adj.12754
23. Demarco GT, Kirschnick LB, Watson LB, Conde MCM, Demarco FF, Chisini LA. What is the clinical applicability of regenerative therapies in dentistry? RGO - Rev Gaúcha Odontol. 2017; 65(4): 359-367. https://doi.org/10.1590/1981-863720170002000113112
24. Bujoli B, Scimeca JC, Verron E. Fibrin as a Multipurpose Physiological Platform for Bone Tissue Engineering and Targeted Delivery of Bioactive Compounds. Pharmaceutics. 2019 Oct 28;11(11):556. https://doi.org/10.3390/pharmaceutics11110556
25. Duncan HF, Kobayashi Y, Shimizu E. Growth Factors and Cell Homing in Dental Tissue Regeneration. Curr Oral Health Rep. 2018 Dec; 5(4): 276-285. https://doi.org/10.1007/s40496-018-0194-y
26. Ozdemir H, Kuzu T. Histomorphometric Evaluation of bone regeneration in peri-implant osseous defects treated with titanium prepared platelet rich fibrin: an experimental study in a rabbit model. Med J SDU / SDÜ Tıp Fak Derg 2018; 25(3): 243-250. https://doi.org/10.17343/sdutfd.332750
27. Andia I, Maffulli N. A contemporary view of platelet-rich plasma therapies: moving toward refined clinical protocols and precise indications. Regen Med. 2018 Sep; 13(6): 717-728. https://doi.org/10.2217/rme-2018-0042
28. Salgado-Peralvo ÁO, Salgado-García Á, Arriba-Fuente L. Nuevas tendencias en regeneración tisular: fibrina rica en plaquetas y leucocitos. Rev Esp Cirug Oral y Maxilofac. 2017; 39(2): 91-98. http://dx.doi.org/10.1016/j.maxilo.2016.03.001
29. Hernández Tejeda N, López Buendía MdC. Elevación de seno maxilar y colocación simultánea de implantes utilizando plasma rico en factores de crecimiento (PRFC), hidroxiapatita y aloinjerto. Reporte de un caso de siete años. Rev Odont Mex. 2013; 17(3):175-180
30. Astuti LA, Hatta M, Oktawati S, Chanda, MH, Djais, AI. Effect of Centrifugation speed and duration of the quantity of platelet rich plasma (PRP). J Int Dent Med Res. 2018; 11(3) 850-857.
31. Dohan DM, Choukroun J, Diss A, Dohan SL, Dohan AJ, Mouhyi J, Gogly B. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part II: platelet-related biologic features. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006 Mar; 101(3): e45-50. http://dx.doi.org/10.1016/j.tripleo.2005.07.009
32. Naik B, Karunakar P, Jayadev M, Marshal VR. Role of Platelet rich fibrin in wound healing: A critical review. J Conserv Dent. 2013 Jul; 16(4): 284-293. https://doi.org/10.4103/0972-0707.114344
33. Bonilla-Sánchez WL, Macio-Pincay JJ, Valarezo-Torres JJ, Medrano-Núñez NV. Plasma rico en plaquetas (PRP) en la cicatrización del tejido mucoso postextraccion. Dom. Cien. 2017; 3(3): 667-83. http://dx.doi.org/10.23857/dom.cien.pocaip.2017.3.3.
34. Anitua E, Prado R, Orive G. Allogeneic Platelet-rich plasma: at the dawn of an off-the-shelf therapy? Trends Biotechnol. 2017 Feb; 35(2): 91-93. http://dx.doi.org/10.1016/j.tibtech.2016.11.001
35. González M, Arteaga-Vizcaíno M, Benito M. Aplicación del plasma rico en plaquetas (PRP) y sus derivados en implantología dental y cirugía plástica. Invest Clin. 2012; 53(4): 408-418.
36. Kobayashi M, Kawase T, Horimizu M, Okuda K, Wolff LF, Yoshie H. A proposed protocol for the standardized preparation of PRF membranes for clinical use. Biologicals. 2012 Sep; 40(5): 323-329. http://dx.doi.org/10.1016/j.biologicals.2012.07.004
37. Uppala S, Parihar AS, Modipalle V, Manual L, Oommen VM, Karadiguddi P, Gupta P. Crestal bone loss around dental implants after implantation of Tricalcium phosphate and Platelet- Rich Plasma: A comparative study. J Family Med Prim Care. 2020 Jan 28; 9(1): 229-234. http://dx.doi.org/10.4103/jfmpc.jfmpc_707_19
38. Acosta Gómez AP, Gutiérrez Prieto SJ, Bedoya Mejía MA, García Robayo DA, Moreno Serrano XE. Evaluación del efecto del plasma rico en plaquetas en diferentes tiempos y concentraciones sobre la viabilidad de fibroblastos de ligamento periodontal y osteoblastos. Univ Odontol. 2017 Jul 28; 36(76). http://dx.doi.org/10.11144/javeriana.uo36-76.eepr
39. Cieslik-Bielecka A, Bold T, ZióBkowski G, Pierchala M, Królikowska A, Reichert P. Antibacterial activity of dental restorative material: An in vitro study. J Conserv Dent. 2018; 21(1): 42-46. https://doi.org/10.1155/2018/9471723
40. Hartshorne J, Gluckman H. A comprehensive clinical review of Platelet Rich Fibrin ( PRF ) and its role in promoting tissue healing and regeneration in dentistry. Part 1: Definition , development , biological characteristics and function. Int Dent. African Ed. 2016; 6(5): 64-69.
41. Meza-Mauricio E, Lecca-Rojas MP, Correa-Quispilaya E, Ríos-Villasis K. Fibrina rica en plaquetas y su aplicación en periodoncia : revisión de literatura. Rev Estomatol Herediana. 2014; 24(4): 287-293.
42. Miron R, Choukroun J, Ghanaati S. Controversies related to scientific report describing g-forces from studies on platelet-rich fibrin: Necessity for standardization of relative centrifugal force values. Int J Growth Factors Stem Cells Dent. 2018; 1(3): 80. https://doi.org/10.4103/GFSC.GFSC_23_18
43. Goel A, Windsor LJ, Gregory RL, Blanchard SB, Hamada Y. Effects of platelet-rich fibrin on human gingival and periodontal ligament fibroblast proliferation from chronic periodontitis versus periodontally healthy subjects. Clin Exp Dent Res. 2021 Aug; 7(4): 436-442. https://doi.org/10.1002/cre2.370
44. Gutiérrez Ramírez DA, Hinojosa Sarria JP, Restrepo Dorado AI, Muñoz Ramírez AL, Velarde Trochez NF, Bastidas Daza FE. Análisis estructural de la fibrina rica en plaquetas y sus aplicaciones en odontología regenerativa. Univ Odontol. 31 de diciembre de 2018; 37(79). https://doi.org/10.11144/javeriana.uo37-79.aefr
45. Dohan Ehrenfest DM, Pinto NR, Pereda A, Jiménez P, Corso MD, Kang BS, Nally M, Lanata N, Wang HL, Quirynen M. The impact of the centrifuge characteristics and centrifugation protocols on the cells, growth factors, and fibrin architecture of a leukocyte- and platelet-rich fibrin (L-PRF) clot and membrane. Platelets. 2018 Mar; 29(2): 171-184. https://doi.org/10.1080/09537104.2017.1293812.
46. Thanasrisuebwong P, Surarit R, Bencharit S, Ruangsawasdi N. Influence of fractionation methods on physical and biological properties of injectable platelet-rich fibrin: An exploratory study. Int J Mol Sci. 2019; 20(7). https://doi.org/10.3390/ijms20071657
47. Durmuş AS, Çeribaşi AO, Can HN. Evaluation of the accelerator effect of coral and platelet rich fibrin on bone healing. Kafkas Univ Vet Fak Derg. 2019; 25(2): 193-199. https://doi.org/10.9775/kvfd.2018.20655
48. Choukroun J, Diss A, Simonpieri A, Girard MO, Schoeffler C, Dohan SL, Dohan AJ, Mouhyi J, Dohan DM. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part IV: clinical effects on tissue healing. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006 Mar; 101(3): e56-60. https://doi.org/10.1016/j.tripleo.2005.07.011
49. Fujioka-Kobayashi M, Miron RJ, Hernandez M, Kandalam U, Zhang Y, Choukroun J. Optimized platelet-rich fibrin with the low-speed concept: growth factor release, biocompatibility, and cellular response. J Periodontol. 2017 Jan; 88(1): 112-121. https://doi.org/10.1902/jop.2016.160443
50. Hartshorne J, Gluckman H. A comprehensive clinical review of Platelet Rich Fibrin (PRF) and its role in promoting tissue healing and regeneration in dentistry. Part II Prep Optim Handl Appl benefits limitations PRF. Int Dent. 2016; 6(5): 34-48.
51. Miron RJ, Moraschini V, Fujioka-Kobayashi M, Zhang Y, Kawase T, Cosgarea R, Jepsen S, Bishara M, Canullo L, Shirakata Y, Gruber R, Ferenc D, Calasans-Maia MD, Wang HL, Sculean A. Use of platelet-rich fibrin for the treatment of periodontal intrabony defects: a systematic review and meta-analysis. Clin Oral Investig. 2021 May; 25(5): 2461-2478. https://doi.org/10.1007/s00784-021-03825-8
52. Mourão CF, Valiense H, Melo ER, Mourão NB, Maia MD. Obtention of injectable platelets rich-fibrin (i-PRF) and its polymerization with bone graft: technical note. Rev Col Bras Cir. 2015 Nov-Dec; 42(6): 421-423. https://doi.org/10.1590/0100-69912015006013
53. Miron RJ, Zucchelli G, Pikos MA, Salama M, Lee S, Guillemette V, Fujioka-Kobayashi M, Bishara M, Zhang Y, Wang HL, Chandad F, Nacopoulos C, Simonpieri A, Aalam AA, Felice P, Sammartino G, Ghanaati S, Hernandez MA, Choukroun J. Use of platelet-rich fibrin in regenerative dentistry: a systematic review. Clin Oral Investig. 2017 Jul; 21(6): 1913-1927. https://doi.org/10.1007/s00784-017-2133-z
54. Inchingolo F, Tatullo M, Marrelli M, Inchingolo AM, Scacco S, Inchingolo AD, Dipalma G, Vermesan D, Abbinante A, Cagiano R. Trial with Platelet-Rich Fibrin and Bio-Oss used as grafting materials in the treatment of the severe maxillar bone atrophy: clinical and radiological evaluations. Eur Rev Med Pharmacol Sci. 2010 Dec; 14(12): 1075-1084.
55. Suparman MK, Wahab HA, Berahim N, Intan T, Jamaluddin BT. Patching up the bone - A case report of autologous fibrin matrix in combination with autogenous bone graft for bone and soft tissue regeneration. Malays Dent J. 2017; 1(1): 1-12.
56. Cortese A, Pantaleo G, Borri A, Caggiano M, Amato M. Platelet-rich fibrin (PRF) in implant dentistry in combination with new bone regenerative technique in elderly patients. Int J Surg Case Rep. 2016; 28: 52-56. https://doi.org/10.1016/j.ijscr.2016.09.022
57. Clem DS III, Lynch SE, Nevins M. Biologics in regeneration: What is their impact on periodontal and dental implant surgery? Compend Contin Educ Dent. 2020 Jan; 41(1): 12-15
58. Hartshorne J, Gluckman H. A comprehensive clinical review of Platelet Rich Fibrin (PRF) and its role in promoting tissue healing and regeneration in dentistry . Part III : Clinical indications of PRF in implant dentistry , periodontology , oral surgery and regenerative endodonti. Int Dent - African Ed. 2016; 6(5): 64-78.
59. Kornsuthisopon C, Pirarat N, Osathanon T, Kalpravidh C. Autologous platelet-rich fibrin stimulates canine periodontal regeneration. Sci Rep. 2020; 10(1): 1-15. https://doi.org/10.1038/s41598-020-58732-x
60. Gasparro R, Sammartino G, Mariniello M, di Lauro AE, Spagnuolo G, Marenzi G. Treatment of periodontal pockets at the distal aspect of mandibular second molar after surgical removal of impacted third molar and application of L-PRF: a split-mouth randomized clinical trial. Quintessence Int. 2020; 51(3): 204-211. https://doi.org/10.3290/j.qi.a43947
61. Príncipe-Delgado Y, Mallma-Medina A, Castro-Rodríguez Y. Efectividad del plasma rico en fibrina y membrana de colágeno en la regeneración ósea guiada. Rev Clín Periodoncia Implantol Rehabil Oral. 2019; 12(2): 63-65 http://dx.doi.org/10.4067/S0719-01072019000200063
62. Yábar-villafuerte G, Becerra-quiñones Y, Obando-pereda GA. Uso del plasma rico en fi brina en endodoncia para regeneración ósea. Reporte de dos casos clínicos. Rev Odontol. Mex. 2018; 22(2): 100-103.
63. Jasmine SAT, Janarthanan K, Krishnamoorthy R, Alshatwi AA. Antimicrobial and antibiofilm potential of injectable platelet rich fibrin-a second-generation platelet concentrate-against biofilm producing oral staphylococcus isolates. Saudi J Biol Sci. 2020 Jan; 27(1): 41-46. https://doi.org/10.1016/j.sjbs.2019.04.012
64. Khorshidi H, Haddadi P, Raoofi S, Badiee P, Dehghani Nazhvani A. Does Adding Silver Nanoparticles to Leukocyte- and Platelet-Rich Fibrin Improve Its Properties? Biomed Res Int. 2018: 8515829. https://doi.org/10.1155/2018/8515829
65. Daugela P, Grimuta V, Sakavicius D, Jonaitis J, Juodzbalys G. Influence of leukocyte- and platelet-rich fibrin (L-PRF) on the outcomes of impacted mandibular third molar removal surgery: A split-mouth randomized clinical trial. Quintessence Int. 2018; 49(5): 377-388. https://doi.org/10.3290/j.qi.a40113
66. Tang YQ, Yeaman MR, Selsted ME. Antimicrobial peptides from human platelets. Infect Immun. 2002; 70(12): 6524-6533. https://doi.org/10.1128/IAI.70.12.6524-6533.2002
67. Canellas JVDS, Medeiros PJD, Figueredo CMDS, Fischer RG, Ritto FG. Platelet-rich fibrin in oral surgical procedures: a systematic review and meta-analysis. Int J Oral Maxillofac Surg. 2019; 48(3): 395-414. https://doi.org/10.1016/j.ijom.2018.07.007
68. He L, Lin Y, Hu X, Zhang Y, Wu H. A comparative study of platelet-rich fibrin (PRF) and platelet-rich plasma (PRP) on the effect of proliferation and differentiation of rat osteoblasts in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009; 108(5): 707-713. https://doi.org/10.1016/j.tripleo.2009.06.044
69. Tsai HC, Chang GR, Fan HC, et al. A mini-pig model for evaluating the efficacy of autologous platelet patches on induced acute full thickness wound healing. BMC Vet Res. 2019; 15(1): 191. https://doi.org/10.1186/s12917-019-1932-7
70. Kushida S, Kakudo N, Morimoto N, et al. Platelet and growth factor concentrations in activated platelet-rich plasma: a comparison of seven commercial separation systems. J Artif Organs. 2014; 17(2): 186-192. https://doi.org/10.1007/s10047-014-0761-5
71. Du Toit J, Siebold A, Dreyer A, Gluckman H. Choukroun. Platelet-Rich Fibrin as an Autogenous Graft Biomaterial in Preimplant Surgery: Results of a Preliminary Randomized, Human Histomorphometric, Split-Mouth Study. Int J Periodontics Restorative Dent. 2016; 36: s75-86. https://doi.org/10.11607/prd.2749
APPENDIX 1
Studies on PRF
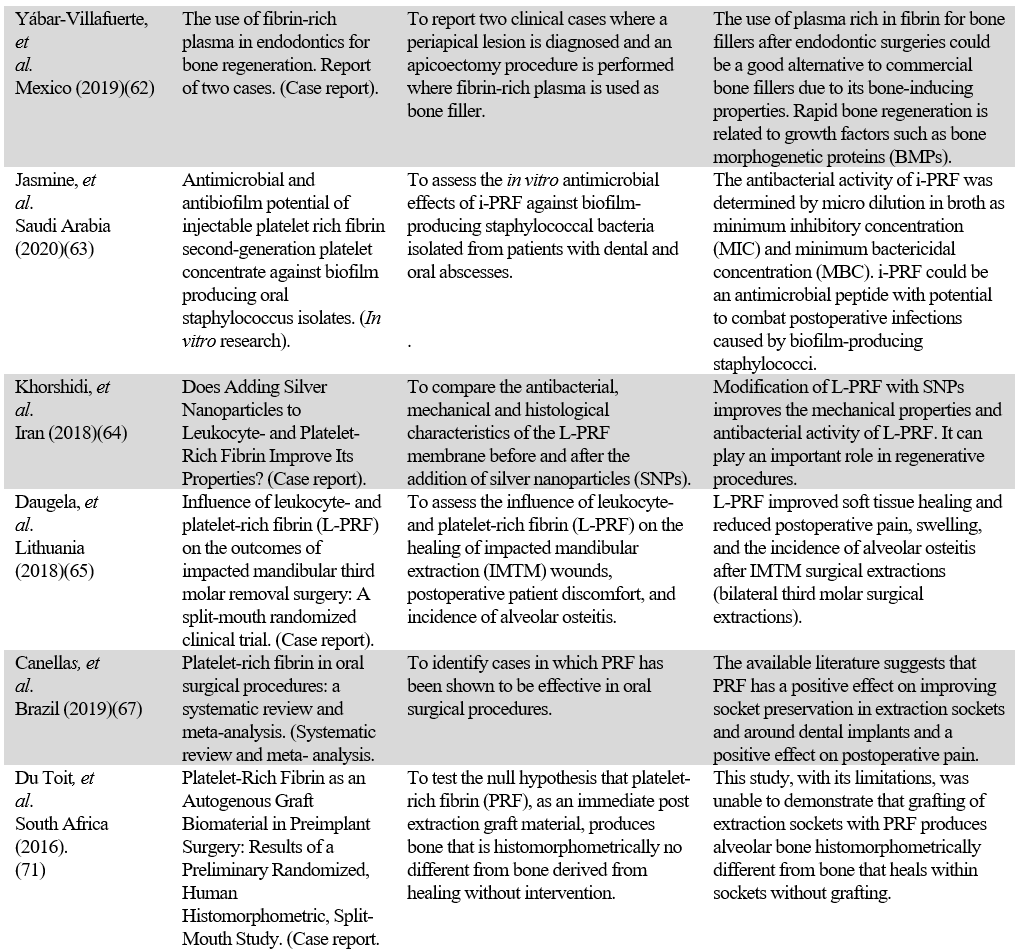
Source: the authors.
Notes
*
Original research.
Author notes
Correspondence: Jc.diazv@javeriana.edu.co; gamboa@javeriana.edu.co
Additional information
How to cite this article: Diaz Velasquez JK, Gamboa Jaimes FO. From Hematology to Tissue Engineering: Current Status and Projection of Platelet Concentrates and their Derivatives. Univ Odontol. 2022; 41. DOI: https://doi.org/10.11144/Javeriana.uo41.fhte